Band Gap Measurement of Titanium Oxide (UV)
Photocatalysts provide catalytic effects when they absorb light and become excited. Because each photocatalyst has a characteristic band gap, they are excited in different wavelength regions. Since UV-visible absorption spectra are used to confirm the excitation wavelengths, they serve as the most fundamental of tools for evaluating photocatalysts. In this example, the band gap of titanium oxide was measured using a UV spectrophotometer. Titanium oxide, which is a material typically used in photocatalysts, has three types of crystal structures - anatase, rutile, and brookite forms. Each type has different density, refractive index, and other properties. The band gap was determined for anatase and rutile type titanium oxide powders, the types mostly commonly used for industrial applications, by measuring the diffuse reflectance.
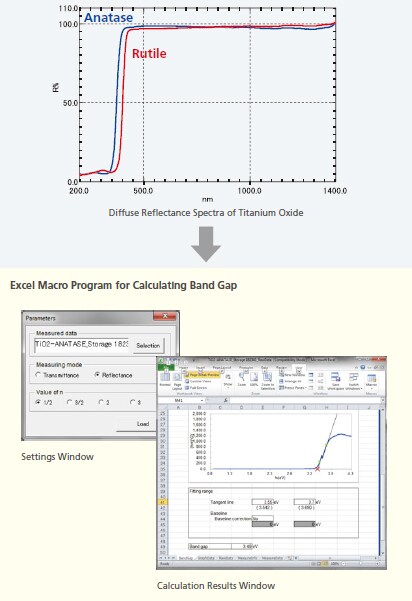
The band gap was determined from the diffuse reflectance spectra. The Excel band gap calculation macro is used by loading spectral data, selecting [Measuring mode (Transmittance/Reflectance)] and [Value of n] (type of transition process), and then specifying the range that can be approximated as a straight line near the inflection point as the tangent line. This makes it easy to determine the band gap value using a Tauc plot.
| Instruments used | UV-2600 UV-VIS Spectrophotometer ISR-2600 Plus Integrating Sphere Attachment |
| Measurement wavelength range | 200 to 1400 nm |
| Scan speed | Medium |
| Sampling pitch | 1.0 nm |
| Photometric value | Reflectance |
| Detector switchover wavelength | 830 nm |
BandGap Calculation Results
| Rutile | 3.20 eV |
| Anatase | 3.49 eV |
UV-2600 UV-VIS Spectrophotometer

- Featuring a compact size and a proprietary Lo-Ray-Ligh grade diffraction grating, the UV-2600 achieves especially high efficiency and low stray light levels.
- The ISR-2600 Plus integrating sphere unit includes two detectors, a photomultiplier tube and InGaAs detector, for high sensitivity measurement capability over a wavelength range from 220 to 1400 nm.
- Combination of the UV-2600 and ISR-2600 Plus, which allows measuring wavelengths ranging from UV to near infrared, is ideal for evaluating the wavelengths absorbed by photocatalysts.


