The Relationship Between UV-VIS Absorption and Structure of Organic Compounds
There are many colored organic compounds, such as dyes and pigments. How is it that these colors come about?
There is a close relationship between the color of an organic compound and its structure. Here, I will explain thisrelationship using absorption spectra of organic compounds obtained with Shimadzu’s UV-2550 UV-VIS spectrophotometer.
1. The Relationship Between Conjugated Double Bond Systems and Absorption Peaks
There are many organic compounds that have conjugated double bond systems (hereafter referred to as “conjugated systems”), in which every other bond is a double bond. These conjugated systems have a large influence on peak wavelengths and absorption intensities.
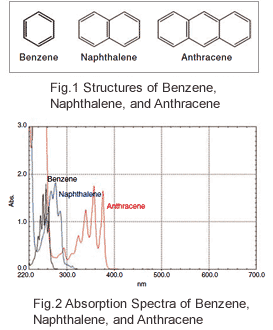
Fig. 1 shows the structures of benzene, naphthalene, and anthracene. Fig. 2 shows the absorption spectra obtained by dissolving these compounds in ethanol and analyzing the resulting solutions. The concentrations were adjusted so that the absorption intensities of the components were roughly the same. It can be seen in Fig. 2 that peak wavelengths tend to be shifted toward the long wavelength region as the conjugated system gets larger. Table 11) gives the peak wavelengths and the molar absorption coefficients of various organic compounds. The molar absorption coefficient is a measurement of how strongly a substance absorbs light. The larger its value, the greater the absorption. With larger conjugated systems, the absorption peak wavelengths tend to be shifted toward the long wavelength region and the absorption peaks tend to be larger.
| Substance | Absorption Peak | Molar Absorption Coefficient |
|---|---|---|
| Ethylene(CH2=CH2) 1.3-butadiene Vitamin A β-carotene |
180nm 217nm 328nm 450nm |
10000 21000 51000 140000 |
| Benzene Naphthalene Anthracene Naphthacene |
255nm 286nm 375nm 477nm |
180 360 7100 110000 |
Table 1 Absorption Peaks and Molar Absorption Coefficients of Various Organic Substances1)
2. Absorption Spectra of Food Dyes with Large Conjugated Systems
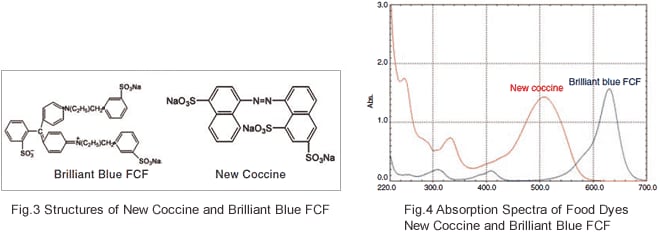
Fig. 3 shows the structures of food dyes New Coccine (Red No. 102) and Brilliant Blue FCF (Blue No. 1) and Fig. 4 shows their absorption spectra. Food dyes tend to have large conjugated systems, like those shown in Fig. 3, and therefore their peak wavelengths tend to be shifted toward the long wavelength region, with peaks appearing in the visible region (400 to 700 nm). This is why they are recognized as colors.
Incidentally, the color that we see is the color that is not absorbed by the substance (which is called the “complementary color”). As shown in Fig. 4, New Coccine absorbs blue and green light in the range 450 to 550 nm, and so the complementary color, red, is seen by the human eye.
Brilliant Blue FCF absorbs yellow light in the range 560 to 650 nm and so blue is seen by the human eye.
3. The Influence of Functional Groups
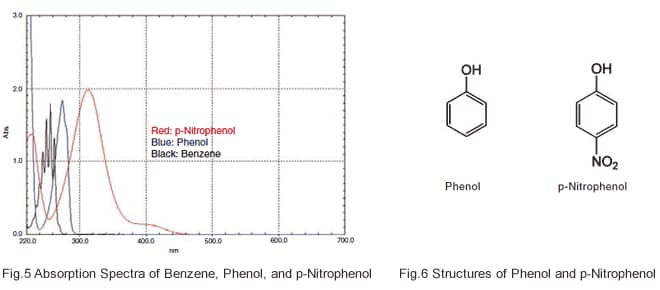
Absorption peaks are also influenced by functional groups. Fig. 5 shows the absorption spectra of benzene, phenol, which consists of a hydroxyl group bonded to a benzene ring, and pnitrophenol,which consists of a hydroxyl group and a nitro group bonded to a benzene ring. The functional groups influence the conjugated systems, causing the absorption peaks to appear at longer wavelengths than the peak wavelength of benzene, although they do not go beyond 400 nm and enter the visible region. The color of organic compounds, then, is influenced more strongly by the size of the conjugated system.
4. Absorption Spectra of Compounds with a Large Molecular Framework and a Small Conjugated System
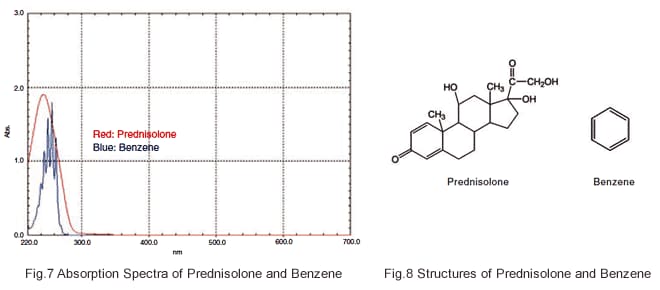
Fig. 7 shows the absorption spectra of prednisolone, which is used as a pharmaceutical, and benzene. Although prednisolone has a large molecular framework, its conjugated system is small and so its peak wavelengths are not shifted greatly toward the long wavelength region, and its peaks appear at roughly the same position as those of benzene.
5. The Reason for the Shift Toward the Long Wavelength Region
I have shown the relationship between molecular structure and absorption spectra. Why, then, does the peak wavelength tend to be shifted toward the long wavelength region as the size of the conjugated system increases? Let us consider the relationship between the energy of light and the movement of electrons.
Light exhibits properties of both waves and particles (photons). The energy of one photon is expressed as hc/λ, where h is Planck’s constant, c is the speed of light, and λ is the wavelength.
Absorption in the ultraviolet and visible regions is related to the transition of electrons. “Transition” refers to the switching of an electron from one state of motion to another. The state of motion of the π electrons in the conjugated system changes more easily than that of the σ electrons that form the molecular frameworks. If a photon collides with a π electron, that π electron readily changes to a different state of motion. This is
true even if the photon has only a small amount of energy. The π electrons in relatively large conjugated systems are more easily affected by low-energy photons. Transition expresses the way that the energy of photons is absorbed by electrons. If a photon has a relatively small amount of energy, the value of hc/λ for that photon is relatively small, and therefore the value of λ is relatively large. λ is observed as the absorption
wavelength and so, if there is a conjugated system, peaks tend to appear in regions where λ is large, i.e., the long wavelength region.
References
1) Masayoshi Nakahara: “The Science of Color”, Baifukan (2002), p. 108


