Particle Size Analysis for Pharmaceuticals

Powders are often used as active pharmaceutical ingredients (APIs) and intermediates of pharmaceuticals. The particle size of APIs and intermediates affects the efficacy of medicines since particle size has an impact on the absorption rate in the gastrointestinal tract after the medicine is taken. It is also important to measure the particle characteristics of raw materials to ensure stable quality production of pharmaceuticals.
Figure 1 shows an example of a pharmaceutical manufacturing process. Chemically synthesized active ingredients are purified and shipped in a powder state. The API is inspected at the delivery destination and then used in the manufacturing of pharmaceutical products. The active pharmaceutical ingredients and additives are weighed, mixed to make the ingredients homogeneous, and then dried to form granules that form a powder.
When manufacturing tablets, granules are compressed into the shape of the tablet. The finished tablets are inspected prior to commercialization, and once confirmed that there are no quality issues, they are sold as products. During this manufacturing flow, particle size is checked at both the time of API delivery and during the manufacturing process.

Fig.1 Flow of Pharmaceutical Manufacturing Process (example for tablets)
Particle Size Analysis for API (Active Pharmaceutical Ingredient) Powder
The high-water solubility of many pharmaceuticals makes wet measurement (dispersing particles in water and measuring their particle size) difficult.
Wet measurements using organic solvents can be applied, but there are issues such as the selection of appropriate dispersing solvents for each type of medicinal product and the cost of purchasing and treating organic solvents. Therefore, dry measurements may be applied to dry powders. A dry measurement unit can measure particle size by dispersing dry powder particles by injecting it into the gas phase prior to a detector.
Figure 2 shows an example of particle size distribution measurement of a dry API powder using the SALD-DS5 dry measurement unit. The API powder’s size used in this example is within the range of 0.5 μm to 80 μm, with a particularly large amount around 20 μm.
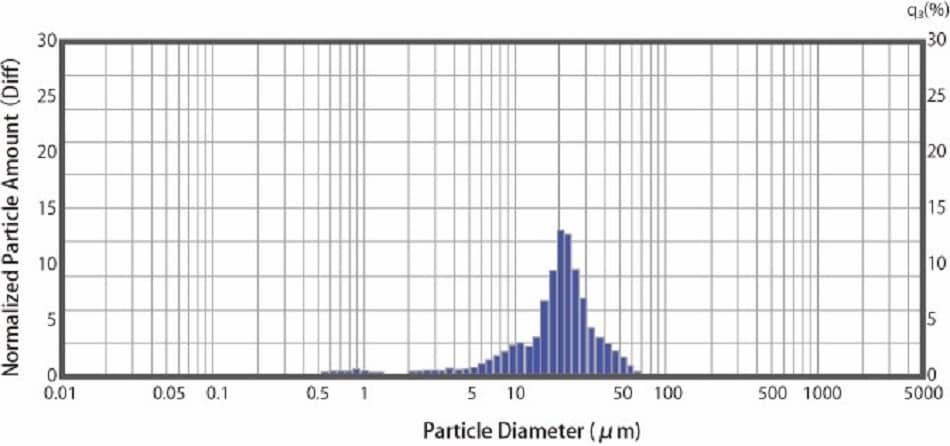
Fig.2 Particle Size Distribution of a Dry API Powder
Measurement of Digestive Medicine Dispersion
Powdered pharmaceuticals such as digestive medicines, consist of primary particles (fine particles) and secondary particles (aggregates), which disperse and turn into primary particles when taken.
Fig. 3 shows the measurement example of the dispersion process of digestive medicine in water using the laser diffraction particle size analyzer : SALD-2300. Data was acquired at 10 second intervals after the medicine was submerged in water. After 10 seconds (black line in Fig. 3 ) there is a large presence of 600 μm particles, which appear to be secondary particles. As time passes, the median size becomes 50-100 μm, while the number of 600 μm particles decreases. This result suggests that the gastrointestinal drug disperses from secondary particles to primary particles.
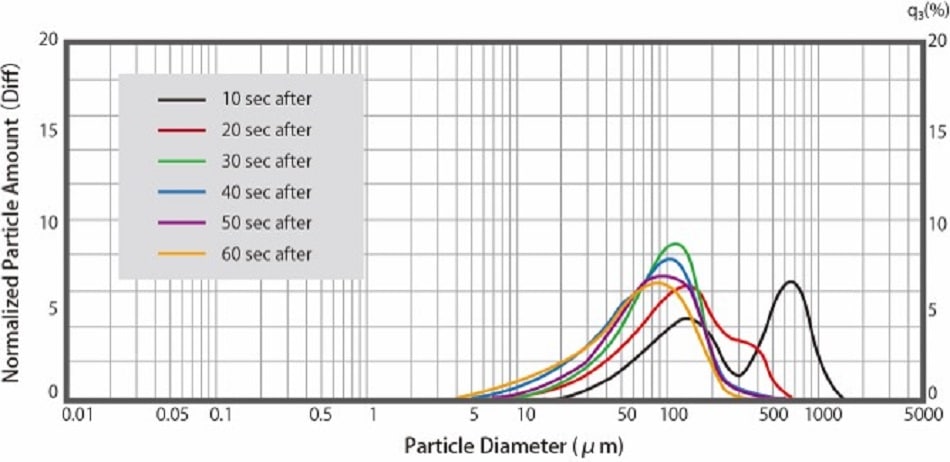
Fig.3 Dispersion of Over-the-counter Gastrointestinal Drugs in Water
Particle Size Analysis of Eye Drop Products
The pharmacopoeia is a standard to ensure the quality of pharmaceutical products. The Japanese Pharmacopoeia has a section on the evaluation of the size and number of insoluble microparticles in eye drops and injectables. Eye drops can be classified as aqueous/nonaqueous eye drops in which the ingredients are dissolved in water or oil, and aqueous/nonaqueous suspension eye drops in which the ingredients are not dissolved, but the particles are suspended. The Japanese Pharmacopoeia states that the maximum particle size of particles in suspension eye drops is 75 μm. Fig. 4 shows a measurement example of a suspension eye drop product using an SALD-2300. The particle size distribution of the eye drops used for the measurement ranged from 0.3 to 20 μm, indicating that the particle size distribution was below the maximum value.
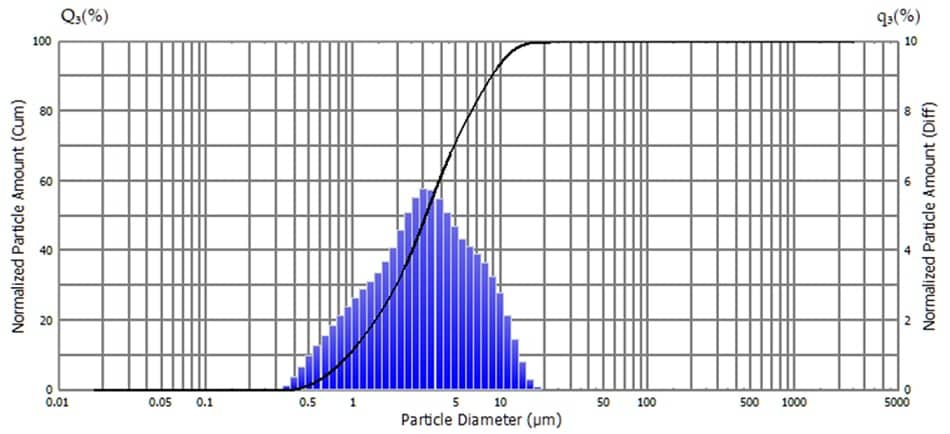
Fig.4 Particle Size Distributions of a Suspension Eye Drop Product
The laser diffraction method is widely used as a technique for measuring particle size distribution because measurement time is short and the measurement range is wide. However, this technique isn’t suitable for measuring the maximum length of aspherical particles because the particle size is calculated by the sphere diameter. The Dynamic Particle Image Analysis System: iSpectTM DIA10 is capable of obtaining information such as the maximum length and size of particles and concentration information from particle images, in addition to particle size distribution. Here, particle size distribution for a suspension eye drop was measured using the iSpectTMDIA10 (Note: the eye drop used for measurement here differs from the one measured by the SALD-2300 above). Fig. 5 shows the measurement result of the suspension eye drop. The thumbnail image (Fig.5 (a)) shows that the particle shape is nearly circular, but each particle has a different shape. Judging from the particle size distribution (Fig.5 (b)), no particles with sizes of approximately 15 μm or larger were detected. The concentration of particles with sizes of 5 μm or larger was exactly 2,652 particles/mL.
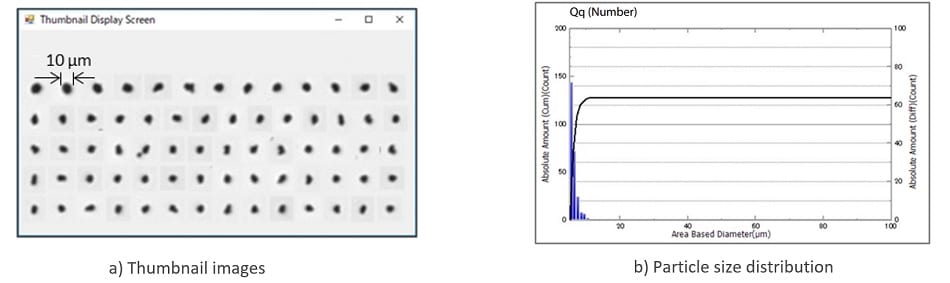
Fig.5 Measurement Results for a Suspension Eye Drop Product
Learn more :
Evaluation of Biopharmaceutical Additive Protein Aggregation Inhibition
A biopharmaceutical (antibody drugs) can exert therapeutic effects by specifically binding to antigens. As a result, it has fewer side effects than a small molecular pharmaceutical and is expected to be highly effective in treating diseases that cannot be treated with small molecular ones. On the other hand, biopharmaceuticals may cause protein aggregation if the solution’s pH or the concentration of additives changes slightly. These phenomenon may not only decrease or eliminate the effect of the drug, but also cause serious side effects such as shock due to an immune response. Therefore, in formulation work for a biopharmaceutical, it is necessary to facilitate conditions where protein aggregation cannot occur. In order to achieve this, investigation of aggregation causes and conditions is necessary. In an experiment, aggregate formation was measured after repeated freezing and thawing of protein solutions with different additives. Seven types of additives were used for this experiment. Those additives were PBS (pH 7.4) and PBS-based solutions adjusted to a pH of 5.8, 6.8, and 7.8, and 0.1% polysorbate 20, 100 mM L-arginine, and 100 mM D-sorbitol in PBS.
Fig. 6 shows the particle size distribution for aggregations in each test solution. Results indicate that aggregate formation is occurring in the 0.2 μm to 10 μm range. Fig. 7 shows the concentration of aggregate formation for each condition. Regarding pH, we can see that lower values correspond to higher concentrations of aggregates, indicating that changing the PBS to a higher acidity increases aggregate concentration. Regarding the polysorbate 20 and L-arginine additives, the intensity of scattered light originating from aggregates was less than the detection sensitivity, meaning almost no aggregates were formed. For D-sorbitol, while aggregate formation was observed, it was inhibited to approximately half when compared to PBS.
This shows that particle size analysis can be used to determine the stability of biopharmaceuticals through the investigation of aggregate formation in formulation solution compositions.
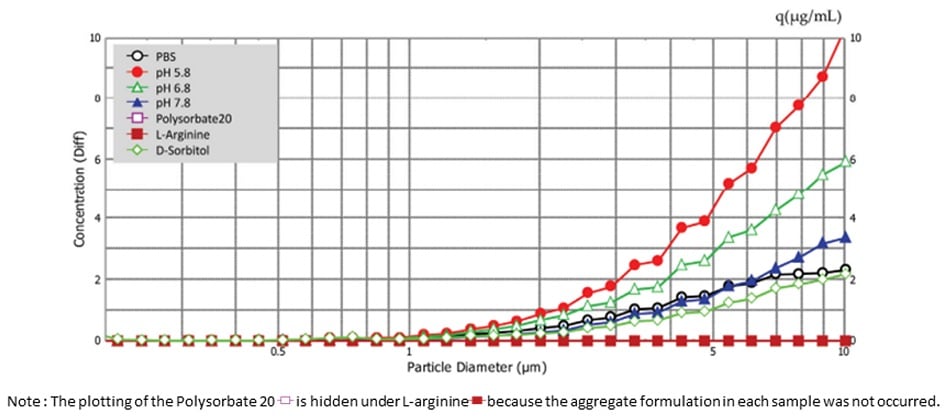
Fig.6 Particle Size Distribution For Aggregation in Each Test Solution
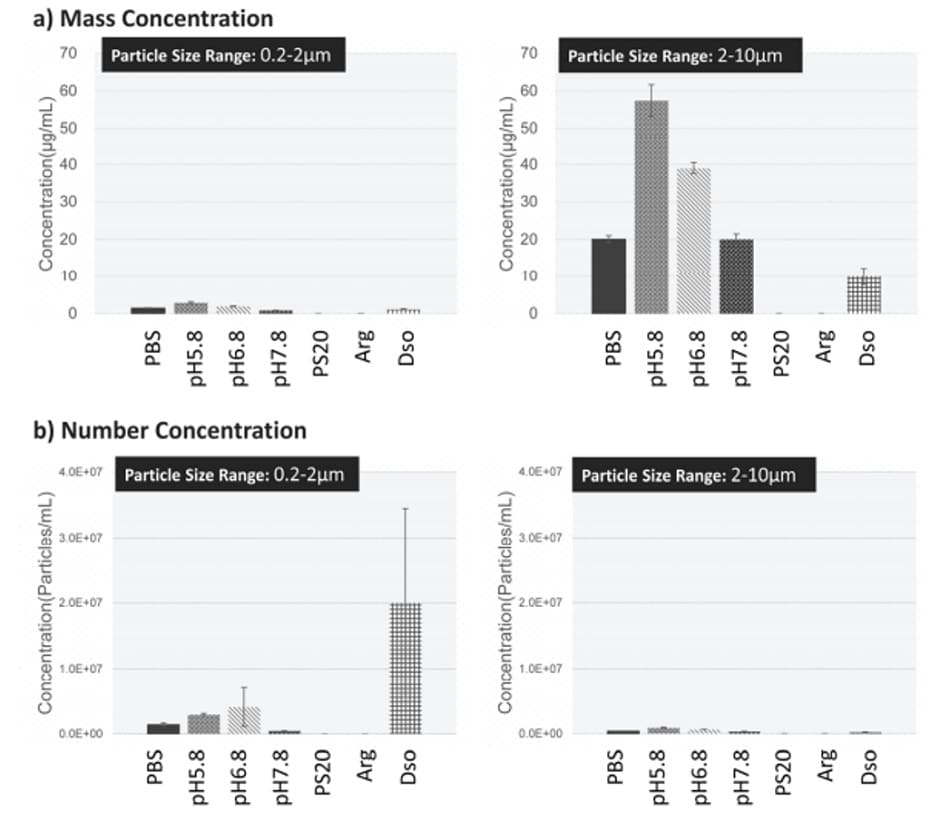
Fig.7 Concentration of Aggregate Formation for Each Condition Learn more : Aggregates Sizer Enables Evaluation of Biopharmaceutical Additive Protein Aggregation Inhibition
Learn more :




