SFC at Preparative Range
GC and HPLC are commonly used to pre-separate samples, but SFC can be used in the same way as well. Preparative separation by SFC offers the following advantages.
- Posttreatment after Preparative Separation
Supercritical carbon dioxide evaporates at ambient temperature and pressure conditions, which eliminates the need for posttreatment. - Solvent Cost for Large Preparative Separation Quantities
Using inexpensive and environmentally-friendly CO2 can reduce the cost of purchasing and disposing of solvents. - Preparative Separation Recovery Rates
After preparative separation, HPLC requires posttreatment steps, such as solvent evaporation or concentration. In contrast, SFC solvents evaporate easily, which minimizes the need for posttreatment and prevents component fragmentation or decomposition during posttreatment.
If SFC is used for preparative separation, an organic solvent that can dissolve target components is sometimes added after column separation to prevent precipitation of target components within flow channels, and gas liquid separators can be used to remove these solvents. A variety of gas-liquid separators are currently being developed by various companies.
Just as with preparative separation by HPLC, SFC involves reviewing results from the analysis scale and then switching to a preparative size column, changing the mobile phase flowrate, and changing the sample injection volume. Equivalent separation can be achieved by increasing the flowrate and injection volume in proportion to the column cross-sectional area. Fig. 1 shows an example of changing from an analysis scale to a preparative scale. By using a column with the same stationary phase, separation equivalent to the analysis scale can be achieved.
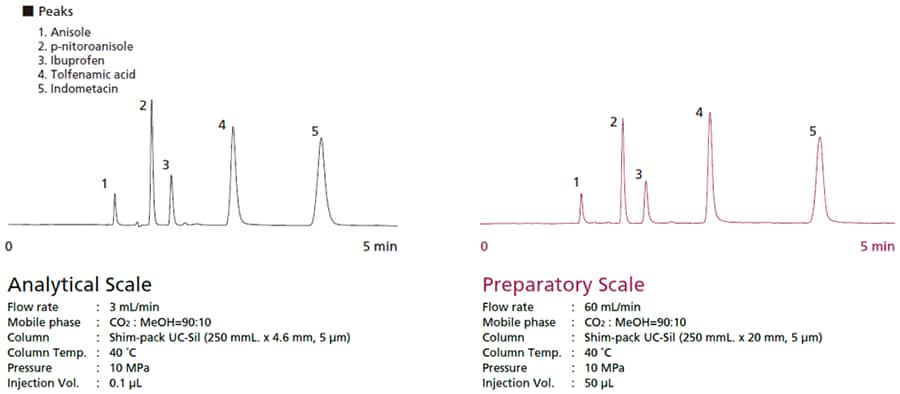
Fig. 1 Migrating from Analysis Scale to Preparative Scale with SFC






