What is Mass Spectrometry?
Shinichi Iwamoto
Mass spectrometry is an analytical technique that is used to measure the mass of molecules. The mass of molecules is almost the same as the molecular weight (MW) that you have learned about in chemistry class.
What can be determined by analyzing air via mass spectrometry?
Using mass spectrometry, the components of air [e.g., nitrogen (N₂, MW=28) and oxygen (O₂, MW=32)] would be detected all at once. Mass spectrometry allows us to determine the mass of molecules; thus, if we detect molecules with the masses of 28 and 32 in air sample, we can easily infer that these masses correspond to N₂ and O₂.
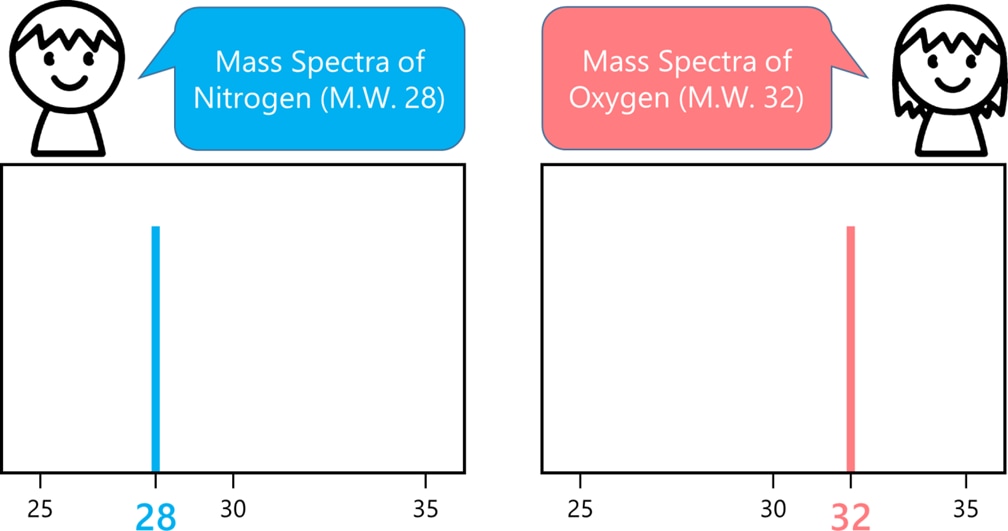
Therefore, mass spectrometry is an excellent technique for analyzing substances, which allows identification of components in a sample by measuring the masses of molecules.
How does mass spectrometry work?
Mass spectrometry consists of three basic steps: “ionization”, “mass separation”, and “detection”.
Ionization is a process by which molecules become charged (charged molecules are called ions). The generated ions have a physical quantity called the mass-to-charge ratio, i.e. the mass of the ion divided by its charge.
During mass separation, ions can be separated according to mass-to-charge ratio in a vacuum vessel by electric or magnetic fields.
Finally, those separated ions arrive at detector and observed as individual signals.
Electric or magnetic fields affect the motion of ions in the vacuum because the ions have electric charges.
The ions are accelerated by applying a voltage and fly in a straight line. As the ions subsequently pass through the magnetic field, they are forced by the magnetic field to bend their orbits. The degree of bending depends on the mass to charge ratio of the ion.
In other words, by precisely setting the electric field or magnetic field, it is possible to determine mass-to-charge ratio of ions of interest or to detect the ions of a specific mass-to-charge ratio.
Of note, the motion of ions in vacuum is solely determined by a physical quantity of mass-to-charge ratio and has nothing to do with the chemistry of the original molecules.
And on to the Nobel Prize
Mass spectrometry originated in England about 100 years ago. In a mass spectrometer used at that time, neon (a noble gas) was ionized by electric discharge in a vacuum vessel. Generated ions were accelerated by the electric potential. Then, ionized neon atoms were separated into ²⁰Ne (mass=20) and its isotope ²²Ne (mass=22) by bending their trajectories by magnetic field. Finally, these ions were detected on a photographic plate.
Currently, mass spectrometers are more sophisticated than they originally were. However, these instruments still consist of three basic components.
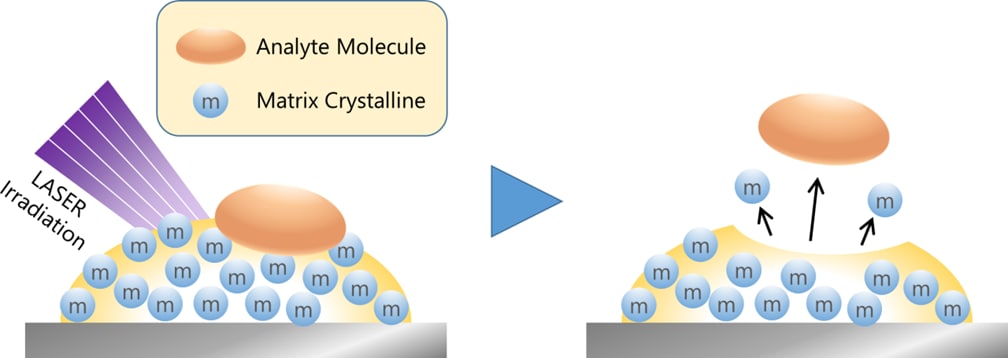
In 2002, John B. Fenn and Koichi Tanaka won the Nobel Prize in Chemistry “for the development of methods for identification and structure analyses of biological macromolecules.” In the 1980s, Fenn and Tanaka developed electrospray ionization (ESI) and soft laser desorption ionization (SLD), respectively, which could be applied to protein analysis. Tanaka's technology led to matrix-assisted laser desorption/ionization (MALDI) mass spectrometry. Mass spectrometer is an indispensable tool for comprehensive protein analysis so-called proteomics, which is widely used in biomarker research. I’d like to highlight that this analysis would not be possible without the advent of ionization technologies such as ESI and MALDI.
Innovative ionization technologies have the potential to considerably expand the scope of analytical science.
Note: This column was originally published on LinkedIn in April 2020. Affiliations and titles are as of that time.
Shinichi Iwamoto, PhD
Deputy General Manager, Koichi Tanaka Mass Spectrometry Research Laboratory
Shinichi Iwamoto joined Shimadzu Corporation in 1991. As a research and development team for biometrics, he developed near-infrared brain function measurement devices and applied research. He was a founding member of the Koichi Tanaka Mass Spectrometry Research Laboratory, established in 2003. Since then, he has been involved in developing elemental technology and applied research of MALDI-MS. He became Deputy General Manager in 2014. He has been engaged in designing fundamental technologies for new mass spectrometers, methods for microbial analysis, and disease biomarkers for cancer and Alzheimer's disease.



