April 18, 2023 | News & Notices
One-Step, Efficient Evaluation of the Thermal Stability of Nucleic Acid Pharmaceuticals
Release of Tm Analysis System, which Achieves the Highest Level of Data Reliability in the Industry

Tm Analysis System
Example of a System with the UV-2600i in the UV-Vis Series of UV-VIS Spectrophotometers
On April 18, Shimadzu has released the Tm analysis system both in Japan and internationally. This analyzes the thermal stability of nucleic acid pharmaceuticals in development using a UV-VIS spectrophotometer. This is the first system to automate the processes from measurement of the Tm value, a standard index of the thermal stability of nucleic acids, to data analysis. The Tm value is indispensable for research and development in the nucleic acid pharmaceutical market, which is expected to have an annual growth rate of approximately 17 % by 2030*.
- *Data source: Cabinet Secretariat, Office of Healthcare Policy “An Investigation of Issues in Pharmaceutical-Related Industrialization and the Initiatives Needed to Solve Them” Reference Materials 3, p.4 (2020)
Nucleic acid pharmaceuticals are constructed using nucleic acids such as DNA and RNA. They have minimal side effects, and their application to a wide range of diseases, including those that are conventionally hard to treat, is anticipated. Accordingly, they are a focus of interest as a third type of pharmaceutical after low molecular pharmaceuticals and antibody pharmaceuticals. In the development and evaluation of nucleic acid pharmaceuticals, confirmation of thermal stability is important, as it is a factor in determining the nucleic acid structure and functionality. When the temperature of nucleic acids is increased, their double strand structure fuses to become a single strand. The temperature at which the ratio of double strand to single strand becomes equal is known as the melting temperature (Tm). This can be analyzed to evaluate the thermal stability of the nucleic acid (Tm analysis). Conventionally, special instruments were required for each measurement procedure, and measurements and data analysis took a great deal of time and effort.
The Tm analysis system automatically performs everything from sample measurements to data analysis in one step. The system consists of a UV-VIS spectrophotometer in the UV-Vis series, the TMSPC-8i 8-series thermoelectrically cooled cell holder, and the LabSolutions UV-Vis Tm measurement analysis software. The newly developed micro cell limits evaporation of the sample, enabling trace quantity measurements, so precious nucleic acid samples are not wasted. The newly designed software is compliant with standards related to data integrity (data integrity concomitant on measurement) as required in the pharmaceutical industry. A procedural log is retained in the database, which can prevent tampering and deletion of data due to procedural mistakes. Users who already have a Shimadzu UV-VIS spectrophotometer in the UV-Vis series can use this system simply by purchasing the cell holder and software.
Shimadzu provides products that support the development of nucleic acid pharmaceuticals. A wide range of products is available, including the MALDI-8030 matrix-assisted laser desorption ionization time-of-flight mass spectrometer, which can measure molecular weights with high accuracy, and the Nexera XS Inert high-performance liquid chromatograph, which can separate and purify target nucleic acids from contaminants. By providing the highly reliable and efficient Tm analysis system, we are contributing to improving the efficiency and reliability of analysis data in the pharmaceutical industry.
Features
1. Enabling Everything from Measurements to Data Analysis in One Step, a First for the Industry
In conventional Tm analysis, multiple processes were required, including measurement of the sample concentrations with a spectrophotometer, creation and configuration of the measurement program, and measurement and analysis of the Tm value with separate software. With our newly designed software, the above-mentioned procedures are implemented automatically in one step. The software automatically calculates the Tm value, preventing data variance due to differences between operators.
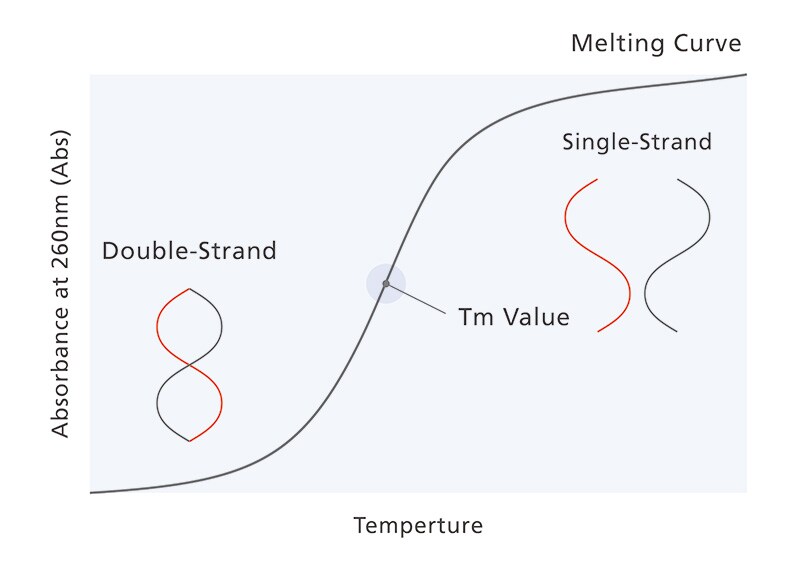
Nucleic acid melting curve. The Tm value refers to the temperature at which the double strand and single strand ratios are equivalent
2. Improves Repeatability and Reduces Costs
Nucleic acid samples used in experiments with nucleic acid pharmaceuticals are obtained in small quantities and are expensive, so reducing the amount used has been an issue. The newly developed micro cell makes it possible to measure the world’s smallest sample volumes while preventing evaporation. This provides low cost data with high repeatability.
3. The Highest Level of Data Reliability in the Industry
In pharmaceuticals evaluation, data integrity and accuracy must be maintained in everything from the drug discovery stage to the analysis results and the concomitant data. The new LabSolutions UV-Vis Tm software achieved the highest data integrity levels in the industry. Measurement-related data including the instrument, operator, measurement results, and audit trail are maintained in the database, preventing tampering and deletion of files by mistake.
- Note 1: UV-1800/1900/2600/2700 series spectrophotometers can be connected to this product.
Note 2: TMSPC and LabSolutions are registered trademarks of Shimadzu Corporation.
For more details, visit
Tm Analysis System


