Principle of Optical Emission Spectrometry
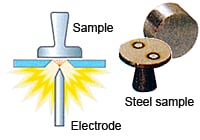
Optical emission spectrometry involves applying electrical energy in the form of spark generated between an electrode and a metal sample, whereby the vaporized atoms are brought to a high energy state within a so-called "discharge plasma".

Spectral lines
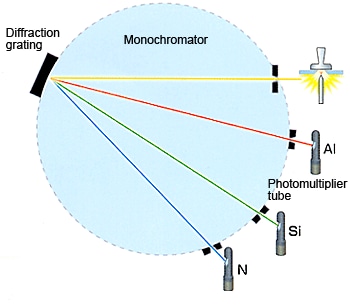
These excited atoms and ions in the discharge plasma create a unique emission spectrum specific to each element, as shown at right. Thus, a single element generates numerous characteristic emission spectral lines.
Therefore, the light generated by the discharge can be said to be a collection of the spectral lines generated by the elements in the sample. This light is split by a diffraction grating to extract the emission spectrum for the target elements. The intensity of each emission spectrum depends on the concentration of the element in the sample. Detectors (photomultiplier tubes) measure the presence or absence or presence of the spectrum extracted for each element and the intensity of the spectrum to perform qualitative and quantitative analysis of the elements.
In the broader sense, optical emission spectrometry includes ICP optical emission spectrometry, which uses an inductively coupled plasma (ICP) as the excitation source. The terms "optical emission spectrometry" and "photoelectric optical emission spectrometry," however, generally refer to optical emission spectrometry using spark discharge, direct-current arc discharge, or glow discharge for generating the excitation discharge.
Shimadzu optical emission spectrometers feature Pulse Distribution Analysis (PDA) to enhance the measurement reproducibility (accuracy). This method involves statistical processing of the spark pulse-generated emission spectra obtained from spark discharges in an argon atmosphere. The optical emission spectrometer offers rapid elemental analysis of solid metal samples, making it indispensable for quality control in steel making and aluminum metallurgy processes.
News / Events
-
Comprehensive ICPMS Maintenance Videos: Keep Your Instruments Performing at Their Best
Our maintenance videos cover everything from detaching and mounting cones to cleaning glassware. These step-by-step guides help ensure your ICPMS performs at its best, preventing issues and extending its lifespan.
-
Energy Dispersive X-ray Fluorescence Spectrometer ALTRACE has been released
Detect trace elements with ease. A combination of optical system design and Shimadzu's proprietary high-speed signal processing technology allows ALTRACE to reach new heights in terms of sensitivity.
-
What is ICP-MS (Inductively Coupled Plasma Mass Spectrometry)?
Basics of Inductively Coupled Plasma Mass Spectrometry is now available.
-
Three Analytical and Measuring Instruments Win the Internationally Recognized “iF Design Award 2024”
The Shimadzu LCMS-2050 high-performance liquid chromatograph mass spectrometer, the Brevis GC-2050 gas chromatograph, and the ICPMS-2040/2050 ICP mass spectrometer won the internationally recognized iF Design Award 2024 in the product design category.
-
New Video: The Cabbage Core, Trash or Treasure?
Most people throw out the cabbage core when cooking, but is the core really just trash or actually a hidden treasure of nutrients?
-
New Video: ICPMS-2040/2050 ProActive Rinsing
ProActive Rinsing can shorten the analysis time by sharting the rinsing sequence in advance. While measuring multiple samples, the rinsing sequence can be started early by sending the autosampler probe to rinse while collecting data using sample already in the suction line. This greatly reduces measurement time and conserves sample.


