

TOC Academy

TOC Measurement Methods
Types of Measurements
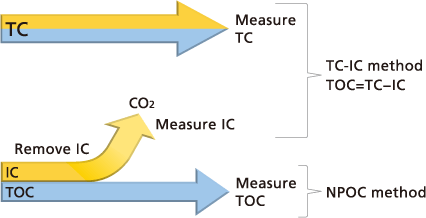
The following two methods are used to determine the TOC present in water.
TC-IC Method:
TOC is determined as the difference between TC and IC measurement values. (TOC = TC - IC)
NPOC Method:
TOC is determined by measuring TC in samples pretreated to remove IC. (TOC = TC)
Measuring IC
TFor TOC measurement, IC refers to the total quantity of inorganic carbon contained inr (where CO2 indicates dissolved carbon dioxide, HCO3- bicarbonate ions, and CO32-carbonate ions). The quantities of dissolved carbon dioxide, bicarbonate ions, and carbonate ions in water are kept in an equilibrium that depends on the pH level of the water, according to the expression below.
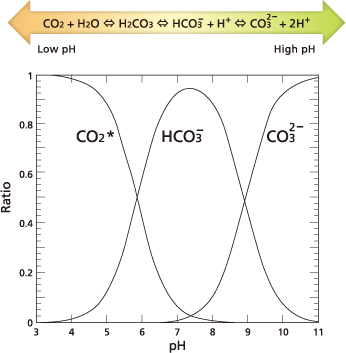
If pH decreases, the equilibrium moves to the left in the expression above. At a pH of 3 or lower, almost all IC becomes dissolved carbon dioxide.
Based on that principle, IC is measured by adding acid to lower the sample pH below 3 and then measuring the CO2 extracted from the sample by bubbling in a CO2-free gas atmosphere.
Using TC-IC and NPOC Methods
Both the TC-IC method and the NPOC method are for measuring TOC, but which of the measurement methods to use is determined based on sample characteristics.
For samples with low IC concentration levels, such as public drinking water or purified water, the NPOC method is used because the TC-IC method is prone to measurement error that could result in lower measurement accuracy.
On the other hand, for samples with large amounts of volatile organic compounds or samples that are prone to foaming, for example, the TC-IC method is used because the NPOC method can result in loss of volatile organic compounds from samples during the bubbling in a CO2-free gas pretreatment step or the NPOC method can prevent accurate sample quantity measurements due to foaming.


