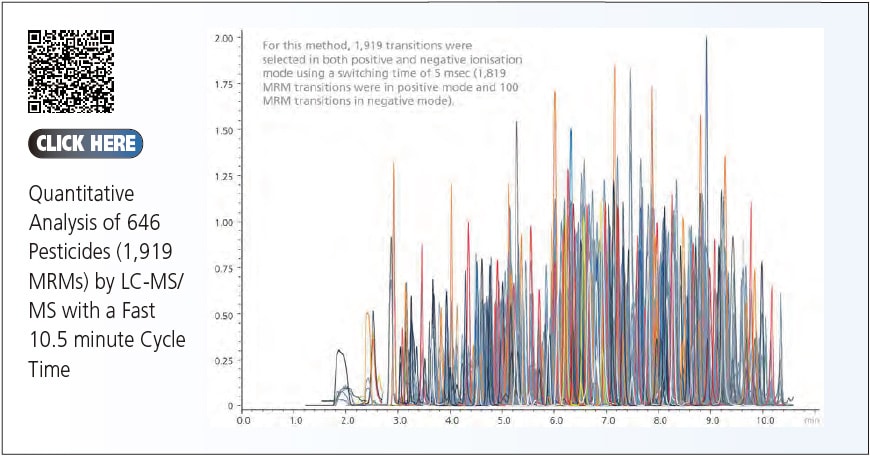
Ultra-Fast Mass Spectrometry (UFMS)
Liquid Chromatograph-Mass Spectrometry
In this section, we shift our focus to Shimadzu's ongoing efforts to overcome these analytical challenges. New technologies, notably Ultra-Fast Mass Spectrometry (UFMS) and Clinical Laboratory Automation Module (CLAM), are developed to increase efficiency and enhance performance of LCMS.
Ultra-Fast Mass Spectrometry (UFMS)
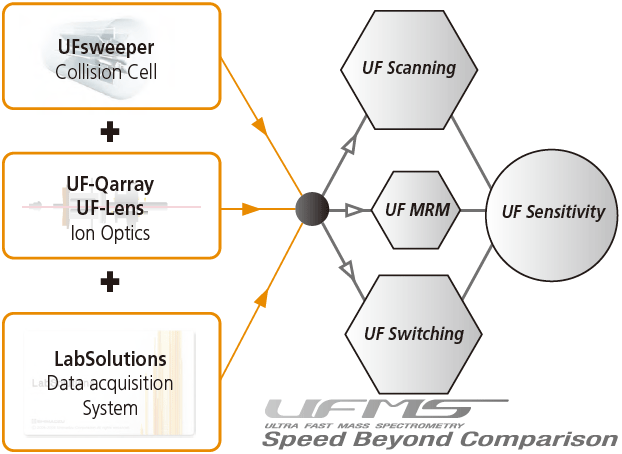
Shimadzu has improved the ion optics, collision cell design and data acquisition systems and developed the UFMS. With the use of UFMS, it can obtain unrivalled sensitivity and performance, that arise due to ultra-fast (UF) scanning, UF switching, UF MRM and UF sensitivity.
Firstly, the ion optics system consists of the UF-Qarray and UF-Lens (Figure 26). With the new design, ions are precisely converged and signal losses are minimized. In addition, the efficiency of the ionization process is improved with the use of a heated ESI probe. Together with the enhanced desolvation line and ion optics system, there is an increase in ion production and transmission, thereby generating a high-intensity and focused ion beam leading to higher sensitivity in LCMS.

Figure 26. Ilustration of the ion source and ion optics (UF-Qarray™ and UF-Lens™) in UFMS.
The next component of the UFMS consists of the UFsweeper. It is a highsensitivity and high-speed collision cell that enables ultra-fast ion sweeping. With the new pseudo potential surface (Figure 27), ions entering the collision cell are accelerated and maintain their momentum upon collision. Under these circumstances, the efficiency of the fragmentation or CID is improved. This technology allows quicker and better ion transmission in the collision cell, maintaining signal intensity and dramatically suppressing crosstalk, even when shorter dwell and pause*9 times are used. Furthermore, this results in the possibility of high-speed MRM.
*9 Measurement conditions must be switched to perform simultaneous measurements of multiple compounds, (multi-component analysis). The time needed for this switching is termed as “pause time”. As data cannot be acquired during the pause time, it should be as short as possible.
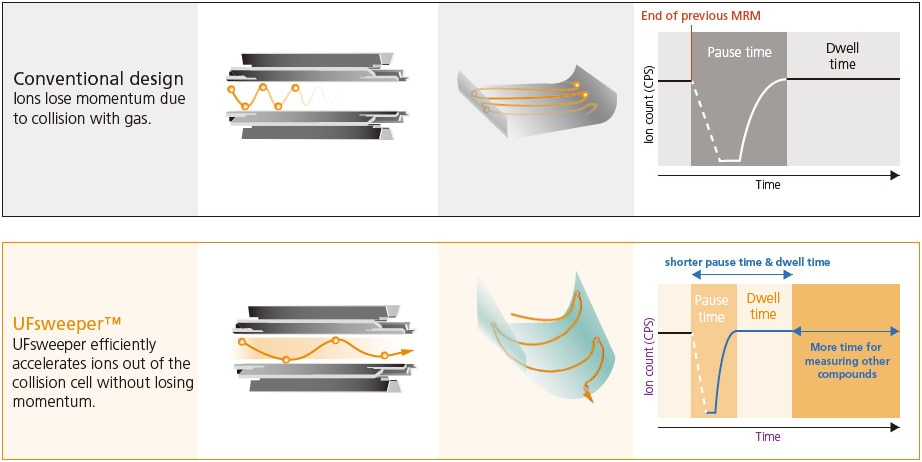
Figure 27. Comparison of a conventional collision cell and UFsweeper™ collision cell.
Together with the developments in data acquisition system, UF polarity switching and UF scanning is made possible. It results in shorter pause time and polarity switching time*10 and bring about more time for data collection. By combining all the UF technologies, the UFMS system delivers UF scanning, switching, sensitivity and MRM to achieve the world's highest levels of sensitivity.
*10 Separate positive ionization and negative ionization modes may be used in LCMS. However, switching between the positive and negative ionizations during analysis is required to and the time taken to switch between these modes is known as the "polarity switching time".
Furthermore, with the use of UFsweeper and high-speed scan analysis in UFMS, simultaneous scan and MRM analysis can be achieved where it alternates rapidly between the scan and MRM modes at high speeds. Both scan and MRM data are obtained from a single analysis. Target compounds can be quantitated from the MRM analysis results while unknown compounds can be identified by comparing the mass spectra to library search results. It maintains resolution and achieves high ion transmission even at high scanning speeds. Therefore, with the use of Shimadzu's UFMS, triple quadrupole MS can be used not only as an excellent instrument for quantitative analysis but also qualitative untargeted analysis.
Clinical Laboratory Automation Module (CLAM)
Besides enhancing the performance and increasing the efficiency of the MS, Shimadzu has looked into innovative solutions that help to simplify the analytical workflow. Pertaining to clinical research, Shimadzu was the first to develop the CLAM, a fully automated sample preparation module for LCMS. From the initial pre-treatment to analysis, it is fully automated and individual samples are analyzed successively in parallel. Clinical scientists working with numerous samples can now utilize this instrument for increased efficiency of existing workflow and improves data reproducibility and accuracy.