Principle of Separation in HPLC
What is the Principle of Separation in HPLC?
High-Performance Liquid Chromatography (HPLC) separates compounds based on how strongly they interact with the mobile and stationary phases. The principle is simple: the more a compound interacts with the stationary phase, the slower it elutes through the column—and vice versa.
How Separation Works in HPLC
There are 3 factors in the column: Compounds, Mobile Phase, and Stationary Phase. Each definition is shown as follows;
- Compounds : Solutes in the sample solution &nbs
- Mobile Phase : Solution delivered using solvent delivery pumps
- Stationary Phase : Functional groups chemically modified in spherical particles packed in the column
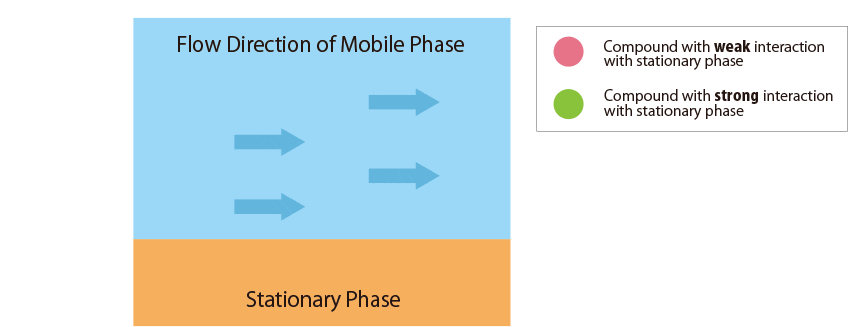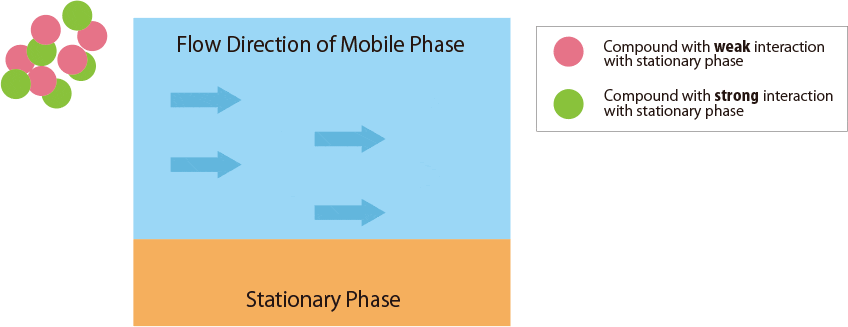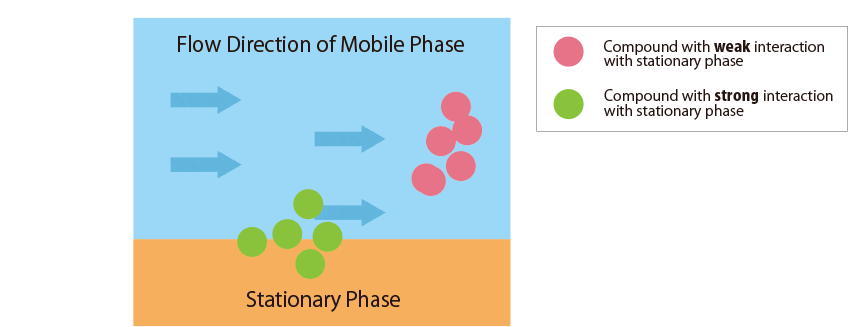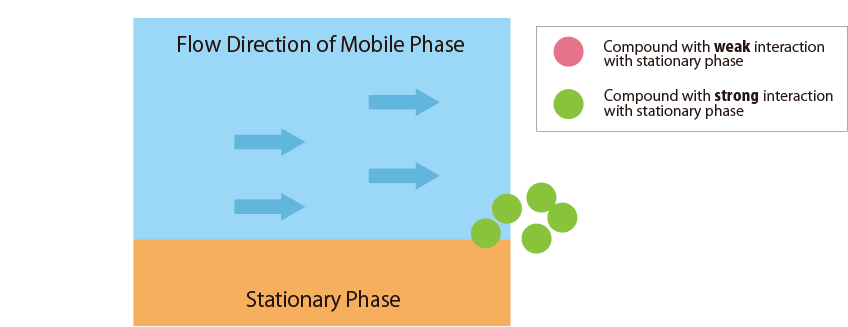
Fig.1 Mechanism of separation in the HPLC column
Fig.1 schematically shows the relationship between the sample compounds and the stationary and mobile phases.
Compounds that interact more strongly with the mobile phase elute more faster from the column.
In contrast, compounds that interact more strongly with the stationary phase remain in the column longer.
"Interaction" refers to the chemical attraction between molecules. The selection of the interaction type suitable for the property of the target compound is necessary for the optimization of the separation method. The type of interaction is related to the separation mode.Table 1 shows the list of general HPLC separation modes and basic principle.
Table 1 List of general HPLC separation modes and basic principle
| Separation Mode | Interaction | Components |
|---|---|---|
| Reversed phase chromatography (RP) | Hydrophobicity(Low Polarity) | Small molecule pharmaceuticals, Vitamins, etc. |
| Normal phase chromatography (NP) | Hydrophilicity (High Polarity) | Saccharides, Nuclear acids, etc. |
| Ion exchange chromatography (IEX) | Electrostaticity | Inorganic ions, Amino acids, Protein, etc. |
| Size exclusion chromatography (SEC) | Molecular size | Synthetic polymer, Biopolymer, Polysaccharide, etc. |
FAQ
Why do some compounds elute faster than others?
Compounds that interact more strongly with the mobile phase elute from the column faster, while compounds that interact more strongly with the stationary phase elute from the column slower.
What is the most common separation mode in HPLC?
Reversed-phase chromatography (RP) is most common, especially for small molecules.
What is the principle of reversed phase HPLC?
The reversed phase HPLC separates compounds based on hydrophobic interactions. The stationary phase is non-polar (typically C18), while the mobile phase is polar. Compounds with more hydrophobicity retain longer in the column, while polar compounds elute faster.
What is the role of the stationary phase in HPLC?
In HPLC, the stationary phase is a packing material inside the column that interacts with the compounds in the sample. The choice of packing material in the column plays a crucial role in determining the types of interactions that influence compound retention and separation during HPLC.






