GC/MS Forensic Toxicological Database
For GC/MS Toxicological Analysis

The GC/MS Forensic Toxicological Database contains information for the simultaneous screening of toxicological substances including drugs of abuse, psychotropic drugs, medical drugs and pesticides. Version 2 adds 1198 compounds to the previous version for a total of 2210 compounds in the database. It also includes optimized methods for headspace analysis of alcohol and volatile toxic substances such as cyanides and azides, providing total support for GC/MS toxicology analysis.
Features
-
The GC/MS scanning method is the gold-standard approach to toxicology analysis, indispensable for the identification of toxicological substances.
-
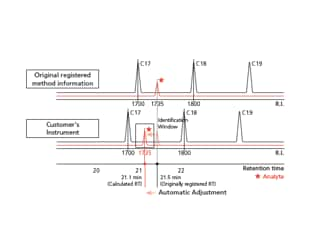
The retention times of all compounds listed in the method file can be simultaneously corrected to your GC-MS system with the Automatic Adjustment of...
-
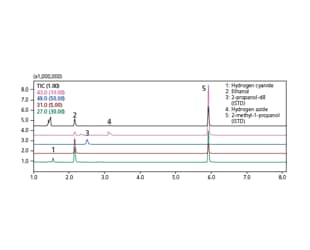
The database provides optimum analysis conditions for blood alcohol and volatile toxic substances, indispensable for forensic police laboratories and...
-
The MS/MS mode of the GCMS-TQ series enables the user to comprehensively detect and estimate the likely structure of cathinones or synthetic...
News / Events
-
Py-Screener Ver.3 has been released
Py-Screener Ver.3 is a screening system for detecting seven types of phthalate esters regulated under the RoHS Directive, brominated flame retardants (PBBs, PBDEs), PIP (3:1) regulated under the U.S. TSCA, UV-328 regulated under the Stockholm Convention on POPs, Dechlorane Plus (DP), and short-chain chlorinated paraffins (SCCPs) / medium-chain chlorinated paraffins (MCCPs).
-
Latest issue of Shimadzu Journal, featuring Environmental Analysis, has come out.
This issue showcases advanced technologies and research tackling the global challenges posed by PFAS. As part of Shimadzu’s ongoing commitment to sustainability and problem solving, we strive to reduce environmental impacts and build a better future.
-
Automatic Derivatization System for Phenethylamine Drugs
This system allows the on-column derivatization using auto-injector AOC-30i and a LabSolutions GCMS system that is now equipped with 2-step injection. On-column derivatization allows for the direct analysis of extracted urine samples without additional sample pretreatment, which can significantly shorten the time required for derivatization.
-
AOAC INTERNATIONAL Posters are now available.
-
Shimadzu has released the GCMS-QP2050
The next-generation GCMS-QP2050 gas chromatograph mass spectrometer, with its accumulation of impressive Shimadzu technology, will lead the way forward. New value is provided by hardware boasting astounding reliability and stability, and easy-to-operate software equipped with superior automated technology.
-
Solutions for Analysis of Ethylene Oxide and 2-Chloroethanol in Food
Even though Ethylene oxide (EtO / EO) is well-known to be a toxic compound with carcinogenic and mutagenic concerns, it has been widely used for fumigation in the food industry because it effectively reduces or eliminates microbiological contamination with bacteria/fungi. The recent recall of food products exported to the EU due to non-compliance with EU regulations has highlighted the importance of quantitation of EtO and 2-CE residues in food.