Triple Quadrupole LC-MS/MS
Solutions for Nitrosamine Analysis -common nitrosamines, NDSRIs using LC-MS/MS, GC-MS/MS and LC-HRMS-
What is Nitrosamine?
Nitrosamines
Nitrosamines are a group of compounds in which the hydrogen of an amine nitrogen is replaced by a nitroso group, and they are a concern due to their potential carcinogenicity. In July 2018, N-nitrosodimethylamine (NDMA) and N-nitrosodiethylamine (NDEA) were detected in the active pharmaceutical ingredient of valsartan, an angiotensin II receptor blocker. Since then, various nitrosamine compounds have been detected in multiple medications, leading to shipment suspensions and voluntary recalls of the drugs, which has become a significant global issue. Regulatory authorities such as the FDA and EMA have issued guidance on the control of nitrosamines in pharmaceuticals, requiring manufacturers to conduct risk assessments and implement risk mitigation measures.
*FDA guidance:https://www.fda.gov/media/141720/download (Refor to FDA)
*EMA guidance:https://www.ema.europa.eu/en/human-regulatory-overview/post-authorisation/pharmacovigilance-post-authorisation/referral-procedures-human-medicines/nitrosamine-impurities (Refer to EMA)
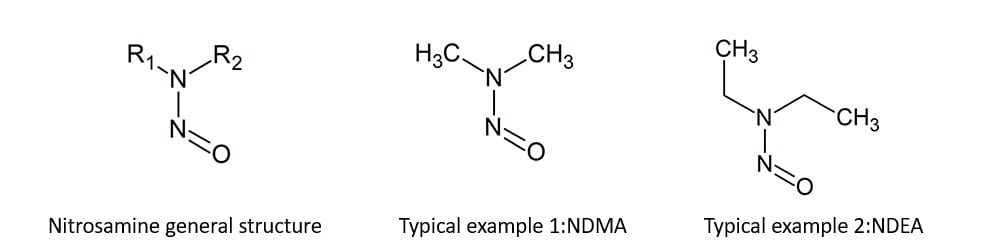
Nitrosamine drug substance-related impurities (NDSRIs)
Nitrosamine drug substance-related impurities (NDSRIs) are a group of compounds in which secondary or tertiary amines in the drug substance are nitrosated. APIs containing secondary or tertiary amines within their molecular structure have been found to react with nitrosating agents to form NDSRI. Recently, it was reported that NTTP, an NDSRI, was detected in the generic medicines containing sitagliptin.
*FDA NDSRI guidance:https://www.fda.gov/media/170794/download (Refer to FDA)

Analysis of Nitrosamines
For the analysis of nitrosamines, highly sensitive and selective methods such as LC-MS/MS, GC-MS/MS, and LC-HRMS are widely used.
USP General Chapter <1469> adopts methods for analyzing nitrosamines in pharmaceuticals using LC-MS/MS and GC-MS/MS.
EP General Chapter 2.5.42 adopts methods for analyzing nitrosamines in pharmaceuticals using GC-MS, LC-MS/MS, and GC-MS/MS.
Read more below to learn how Shimadzu can meet your nitrosamine analysis needs including NDSRI, from routine lot and batch testing to present and future drug discovery.
Applications
User Benefit
- Meet Japanese, U.S., and European risk assessment standards for contamination with nitrosamines.
- Reliable quantitative analysis of nitrosamines in an active pharmaceutical ingredient.
User Benefit
- Simple and sensitive LC-MS/MS method to quantify ten nitrosamine impurities with simple sample pre-treatment
- Achieve good linearity of R2> 0.999 with recovery within 70 – 120 % without matrix-matched calibration
User Benefit
- This method exhibits high sensitivity with a quantitation limit of 5 ng/day, significantly surpassing the acceptable intake limit of 37 ng/day.
- The pretreatment process for this method is straightforward and easy to perform, requiring only ultrasonication.
User Benefit
- Dynamic headspace equipped with GC-MS/MS was used for trace level quantitation of Nitrosamines in Metformin API
- GCMS-TQ8050 NX system equipped with HS-20 easily meets the criteria as per the regulatory guidelines on Nitrosamines
- Compared to static headspace, dynamic headspace has advantage in trace level detection of Nitrosamines
User Benefit
- A HS-GCMS/MS method for the determination of 5 Nitrosamines in Metformin API & formulation
- The GCMS-TQ8040 NX system easily meets the criteria as per the USFDA guidance on Nitrosamines
User Benefit
- Simultaneous analysis of up to eight N-nitrosamine impurities in drug substances by LC-HRMS method on LCMS-9030.
- A targeted MS/MS (TOF) method with 2 m/z isolation window by the quadrupole was optimized to obtain best sensitivity.
- A mass tolerance of (±)15 ppm was adopted to produce extracted-ion chromatograms (XICs) for quantitation.
-
-
Triple Quadrupole Gas Chromatograph Mass Spectrometer
-
Triple Quadrupole Gas Chromatograph Mass Spectrometer
-
Quadrupole Time-of-Flight Liquid Chromatograph Mass Spectrometer







