Changing the Carrier Gas
- Measures and Proposals to Reduce Helium Gas Consumption
- Reducing Carrier Gas Consumption
- Changing the Carrier Gas
The following explains precautions and other information for switching the carrier gas from helium to hydrogen or nitrogen.
Changing the Carrier Gas to Hydrogen
Helium is normally used as the carrier gas for GCMS analysis. However, helium cannot be used in some situations; for those analyses, hydrogen can be used. Shimadzu's gas chromatograph mass spectrometers (GCMS) are compatible with hydrogen carrier gas, allowing hydrogen to be used as a carrier gas for the GC unit. Hydrogen gas (H2) is easier to obtain and less expensive than helium gas, and can maintain high separation performance for a given linear velocity. If used safely, it can also significantly reduce running costs.
| * | If the type of carrier gas is changed, it may not be possible to achieve the same results using the same analytical conditions. Therefore, it may require reviewing the specified analytical conditions. |
| * | GCMS pretreatment systems use other gases beside carrier gas, which might prevent using hydrogen as a carrier gas, depending on the type of system involved. For information about pretreatment systems that support using hydrogen as a carrier gas, contact a Shimadzu sales representative. |
GCMS-TQ series, GCMS-QP2020 series, and GCMS-QP2010 series models include a carrier gas automatic shutdown function. An electronic flow controller (AFC), used to control the carrier gas flowrate, shuts off the carrier gas supply and automatically stops the GC unit if the column inlet pressure or the total flowrate does not reach the corresponding setting value within given parameters, assuming a major carrier gas leak or other problem has occurred. The carrier gas supply is also shut off automatically if the vacuum pump stops due to a failure.
Features for using hydrogen as a carrier gas are listed below. Verification of using hydrogen as a carrier gas in GCMS-TQ series and GCMS-QP2010 series systems is also described.
- By monitoring the pressure and flowrate at the injection port, the oven temperature is decreased, and carrier gas supply is shut off if the pressure and flowrate do not reach specified values within a certain time.
- If a carrier gas control problem occurs, the electronic flow controller automatically shuts off the carrier gas supply.
- If the vacuum pump stops due to a failure, the carrier gas supply is automatically shut off.
- Shimadzu GCMS systems are tested for hydrogen leaks inside the oven to confirm that the oven design does not permit hydrogen to collect inside the oven. Due to the high diffusion coefficient of hydrogen, it will not collect inside the oven.
- Shimadzu GCMS systems are explosion tested using hydrogen. Given analytical conditions normally used, hydrogen gas only causes a small explosion sound when intentionally ignited inside the column oven. Also, after a vacuum is pulled, fully loosening the knob on front door of the MS unit can prevent hydrogen from collecting inside the MS unit, under normal operating conditions.
- Shimadzu GCMSsolution workstation software can be configured with hydrogen as the standard carrier gas and migrated methods can be reviewed easily by specifying the constant carrier gas linear velocity mode.
Be sure to do the following when using hydrogen gas.
| If a leak error occurs using an electronic flow controller and APC/AFC unit, even though the gas supply pressure is normal, stop using the system and request service from a Shimadzu representative. If no leaks are found, leakage does not stop, or normal performance is not restored after stopping the leak, stop using the system and request service from a Shimadzu representative. |
| Fully loosen the front door knob on the MS unit after a vacuum has been pulled. |
Precautions for Using (Handling) Hydrogen Carrier Gas Safely
Hydrogen is a dangerous gas that can easily explode. A Safety Manual was prepared that lists the possible risks associated with using hydrogen gas in a gas chromatograph mass spectrometer and indicates considerations for ensuring the systems are used safely.
>> Using hydrogen carrier gas safely in GCMS-QP2010 series, GCMS-QP2020, and GCMS-TQ8030/8040 systems ![]() (228 KB)
(228 KB)
Considerations That Could Affect Analysis
It will increase noise in terms of sensitivity (S/N). For S/N values using hydrogen gas in respective systems, refer to product specifications. Equivalent separation results can be obtained by using an optimal column and creating a method. Most compounds can be analyzed, but hydrogen is not an inert gas, so it can dissociate at the injection port in some cases.
Changing the Carrier Gas to Nitrogen
Carefully review the condition settings before switching the carrier gas to nitrogen.
Nitrogen gas (N2) is an inexpensive and safe gas. However, if nitrogen gas is used in a capillary GC system with the same analytical condition settings as for helium, then separation capabilities will decrease in most cases. If there are no adjacent peaks involved, then the carrier gas can be changed to nitrogen without changing the condition settings. If there are many adjacent component peaks and a certain level of separation is required, then the condition settings need to be reconsidered before using nitrogen gas.
Improving separation capabilities for nitrogen gas will require reconsidering the carrier gas linear velocity and temperature settings and will probably result in a longer analysis time. The vaporization state and detector sensitivity will probability change and the peak area percent might differ from when helium gas was used.
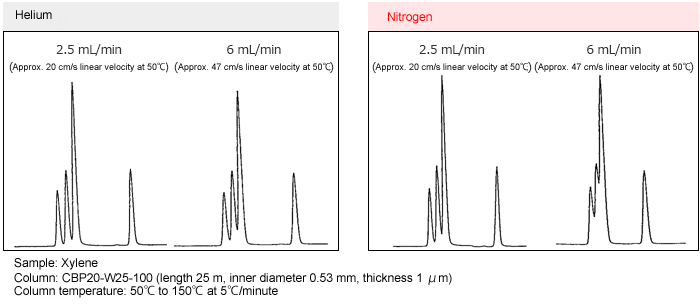
Separation with Helium Gas and Nitrogen Gas
Separation examples for helium and nitrogen gases at different linear velocities are shown below.
Using helium gas, the separation is virtually unchanged across the range from 20 to 47 cm/s linear velocity. With nitrogen gas, however, the separation deteriorates at 47 cm/s linear velocity. This occurs because the optimal linear velocity for separation is lower with nitrogen than with helium and the optimal linear velocity range for nitrogen is narrower than for helium.
Reviewing Analytical Conditions for Nitrogen Gas
If samples are analyzed with nitrogen gas using the same conditions as optimized for helium gas, then the separation capability will decrease due to different column efficiency (HETP). If high separation is unnecessary, then the same analytical conditions can be used in some cases. If high separation is required, however, it is recommended that analytical conditions be reconsidered, starting at a linear velocity in the 10 to 20 cm/s range where separation performance tends to be higher. (Slower linear velocity values will result in longer analysis times.)
That means the linear velocity will vary as the temperature increases when performing programmed temperature analysis, but Shimadzu GC units use constant linear velocity control functionality to provide optimal separation even when using nitrogen gas.
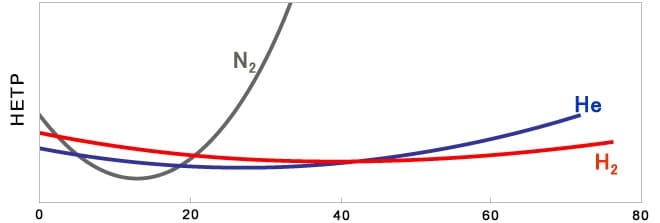
Average linear velocity (cm/sec)


