PPSQ-51A/53A
Protein Sequencer
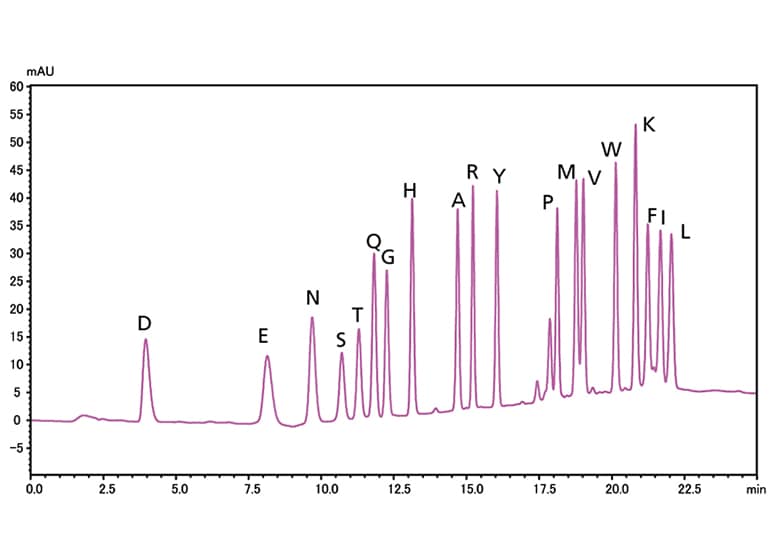
The PPSQ is an instrument for determining the amino acid sequences of proteins and peptides, which combines an Edman reaction section with a high performance liquid chromatograph (HPLC). There are 2 types: the PPSQ-51A, which is equipped with one reactor, and the PPSQ-53A, which is equipped with three reactors. On the PPSQ-53A, the continuous analysis of the amino acid sequences of multiple samples can be performed one after another. In the Edman reaction section, amino acids are cleaved in order from the N-terminal of a protein by repeatedly performing Edman degradation, and are derivatized. As a result, stable PTH-amino acids are produced. The PTH-amino acids are injected online into the HPLC, and analysis is performed. The HPLC data is saved on the PC, and data processing software is used to process the chromatograms. Then, amino acid sequence estimation software is used to identify the amino acids and estimate the sequences.
Benefits of using Edman degradation for amino acid sequencing
(1) Guaranteed N-terminal sequence of proteins
(2) No sample pre-treatment required
(3) Differentiation of isobaric amino acids (leucine and isoleucine)
(4) Identification possible even from unknown proteins not registered in databases
Features
-
LabSolutions PPSQ software provides compliance with FDA 21 CFR Part 11 guidelines and enables compliance with the security, user management, and audit trail requirements specified by FDA 21 CFR Part 11....
-
Baseline Stability
PPSQ series protein sequencers separate PTH-amino acids isocratically. This improves baseline stability and allows high ... -
High-Sensitivity Analysis
A high-sensitivity flow cell enables high-sensitivity detection of PTH-amino acids, which allows for sequential analysis using trace samples...
News / Events
-
New IMAGEREVEAL MS, Mass Spectrometry Imaging Data Analysis Software
Automatically uncover important information from large amounts of data with IMAGEREVEAL MS.
-
New iMScope QT
Inheriting the concept of a mass spectrometer equipped with an optical microscope from the iMScope series, the iMScope QT is also Shimadzu's flagship model for MS imaging with a LCMS-Q-TOF.
-
Shimadzu has released the Cell Pocket Ver.2.10 Web Application Supporting Cellular Observations
Cell Pocket is an integrated system for both the analysis and management of cell images.
-
Shimadzu has released the Tm Analysis System
Tm analysis systems can accelerate the development process and improve the quality of oligonucleotide therapeutics. Control by LabSolutionsTM software enables compliance with ER/ES-related regulatory requirements and improves the efficiency of analyzing the thermal stability (Tm analysis) of nucleic acids.
-
Dementia / Alzheimer‘s disease
Shimadzu is a leading driver in developing healthcare solutions with technology developments ranging from advanced imaging systems for use in medical diagnosis and treatment, engineering mass spectrometry-based solutions for exploring ways to discover disease biomarkers as well as building MS solutions for daily routine quantitation in biological fluids.
-
Shimadzu has released Cell Pocket Ver.2
Cell Pocket is an integrated system for both the analysis and management of cell images.