Effects of Sample Solvents on Peak Shape
The sample injected by the autosampler reaches the separation column through the tubing. If the mixture of sample solvent and mobile phase is insufficient before the sample solvent reaches the column, the peak shape may deteriorate depending on the type of sample solvent. The peak shape may be broadened when using a sample solvent with a higher elution intensity than the mobile phase. Additionally, the use of the UHPLC system which employs tubing with smaller inner diameter is prone to be affected with inadequate mixing of the sample solvent and mobile phase.
Fig. 1 shows the effect of the elution strength of the sample solvent on the peak shape, using the theoretical plate number during caffeine analysis by reverse phase chromatography as an example. The parameter of peak shape is represented by the theoretical plate number (TPN), which it indicates the sharpness of the peak. In this case, the mixture of methanol / water (3:7) is used for the mobile phase. Comparing TPNs when using methanol as the sample solvent, which has a higher elution strength than the mobile phase, and water, which has a lower, you can see that there is a difference in TPNs. When the injection volume is further increased, you can see there is a remarkable difference Fig. 2 shows a comparison of the injection volume and peak shape when methanol is used as the sample solvent. The sample concentration is adjusted so that the absolute injection amount is the same. It can be seen that the peak shape deteriorates as the injection volume increases.
-
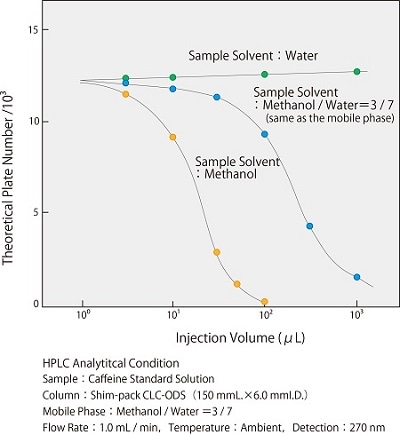
Fig.1 Effect of Elution Strength of Sample Solvent on TPNcaption -
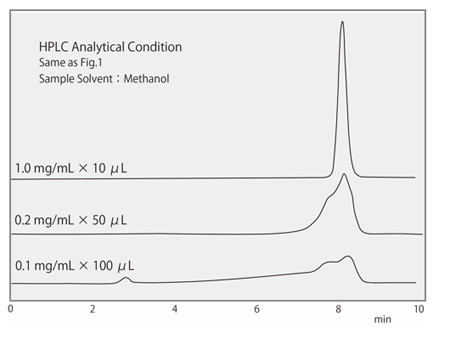
Fig.2 Comparison of Peak Shape Between Injection Volume
Fig.3 describes an image of the analytes and the sample solvents moving through the column. Sample solvent can be considered as part of the mobile phase in the column but using a sample solvent with higher elution strength may move the analytes faster, as if it were a mobile phase. In contrast, as for the use of a sample solvent with lower elution strength, the analyte initially concentrates then once more starts to separate in the mobile phase because the sample solvent acts as a mobile phase with lower elution strength. Using a sample solvent with an elution strength lower than that of the mobile phase can sharpen the peak shape because the diffusion of the analyte can be suppressed in the column.
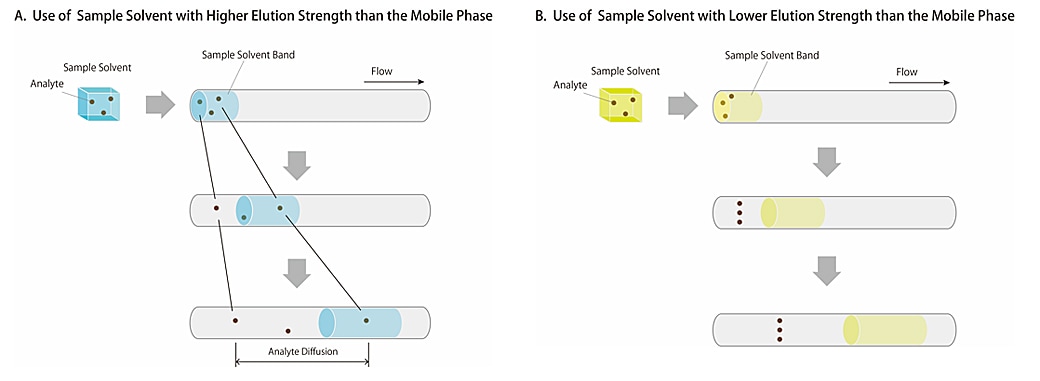
Fig.3 Comparison of the retention by using 2 types of sample solvent with different elution strength
As mentioned above, it is explained that the difference in the elution strength between the sample solvent and the mobile phase affects the peak shape. It can be sharpened by using a sample solvent with a lower elution strength than the mobile phase, but if the sample is prepared with a solvent with a higher elution strength (e.g.; methanol 100% in RP-LC), there is concern about the trouble of further pretreatment such as substitution and dilution.
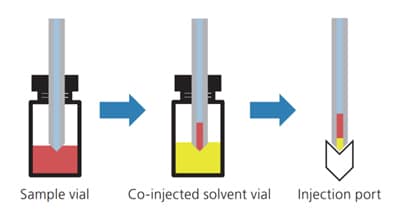
Fig.4 Image of the Injection Process
Autosamplers in the Nexera Series(LC-40 Series) and i-Series are equipped as standard with a variety of automatic pretreatment functions such as the co-injection function. Fig. 4 describes the process of operation in the co-injection mode. The use of this mode allows the solvent for co-injection to be automatically aspirated from a specific vial and injected with the sample into the analytical column. Therefore, a sharp peak can be obtained without the trouble of manually replacing or diluting. Fig.5 shows a comparison of chromatograms with and without co-injecting dilution solvent (water) in the analysis of samples prepared with a solvent that has a higher elution strength than the mobile phase in reversed-phase analysis. This comparison is the result of analysis by gradually increasing the injection volume of the diluent. Chromatograms obtained with co-injection show that increased injection volume does not contribute to peak shape, resulting in sharp peaks in all analyzes.
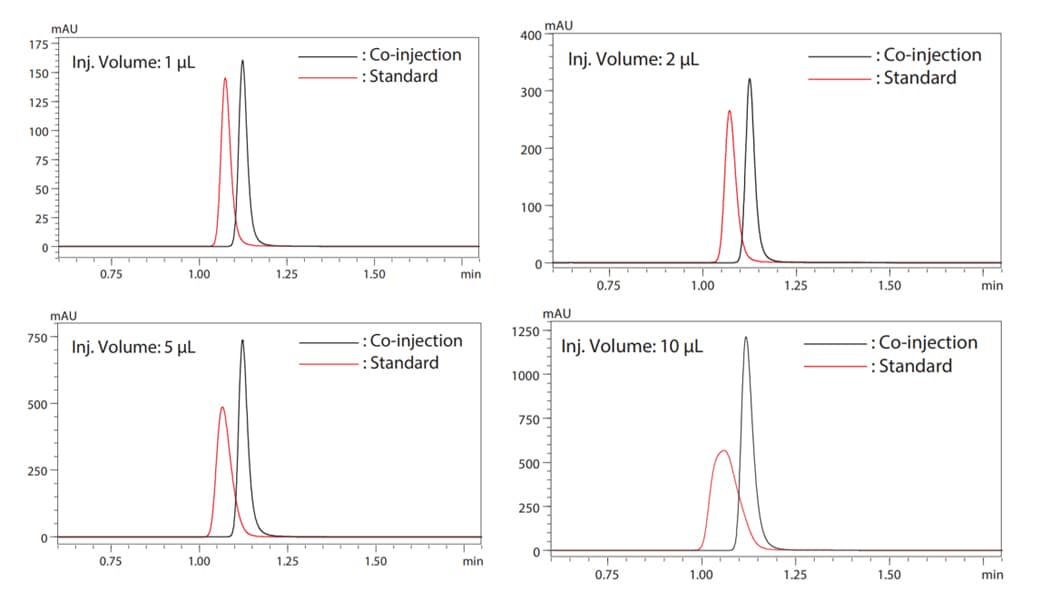
Fig.5 Peak Shape Comparison When Diluent is Co-injected Peak Shape Comparison When Diluent is Co-injected
In the analysis of the residues of pesticides and veterinary drugs, only organic solvent, 100% methanol, may be replaced with the sample solvent in preparation for solid-phase extraction. In particular, when the analytes are highly polar compounds in the reverse phase chromatography, a combination of a high ratio of water as a mobile phase and a sample solvent having a higher elution strength tends to cause deterioration of the peak shape. Co-injection with the automatic pretreatment function realizes analysis that guarantees quantitative reliability ands obtain the good peak shape.
Links


