Tips for LC and LC/MS Analysis (Vial Selection): Interactions to Be Aware of When Using Vials
1. Leaching from vials
Leaching from sample vials can affect analytical results in three primary ways:
- Leachables from vial components being detected as impurity peaks.
- In LC/MS analysis, metal leached from vial components being detected as adducts of the target analytes.
- Time-dependent metal leaching from vial components can alter sample solvent pH, potentially promoting degradation of the analytes.
Here, we present examples using glass vials as a representative vial type. Vials are primarily made of borosilicate glass. Borosilicate glass contains metal oxides added during manufacture to improve formability*1. These metal species are known to be concentrated at the glass vial surface*2. Metals on the inner vial surface can cleave siloxane bonds and form metal silanolates. Metal silanolates are readily ionized when sample solvent is added to the vial*2,3, so leached metal ions can affect analytical results*4. When a neutral sample solvent is dispensed into the vial, metal silanolates dissociate and representative alkali metals present in borosilicate glass—such as sodium ions—are released. As a result, leaching of Na+ can raise the solvent pH, potentially promoting degradation of the analytes or altering their solubility. The schematic below illustrates this process (Figure 1).
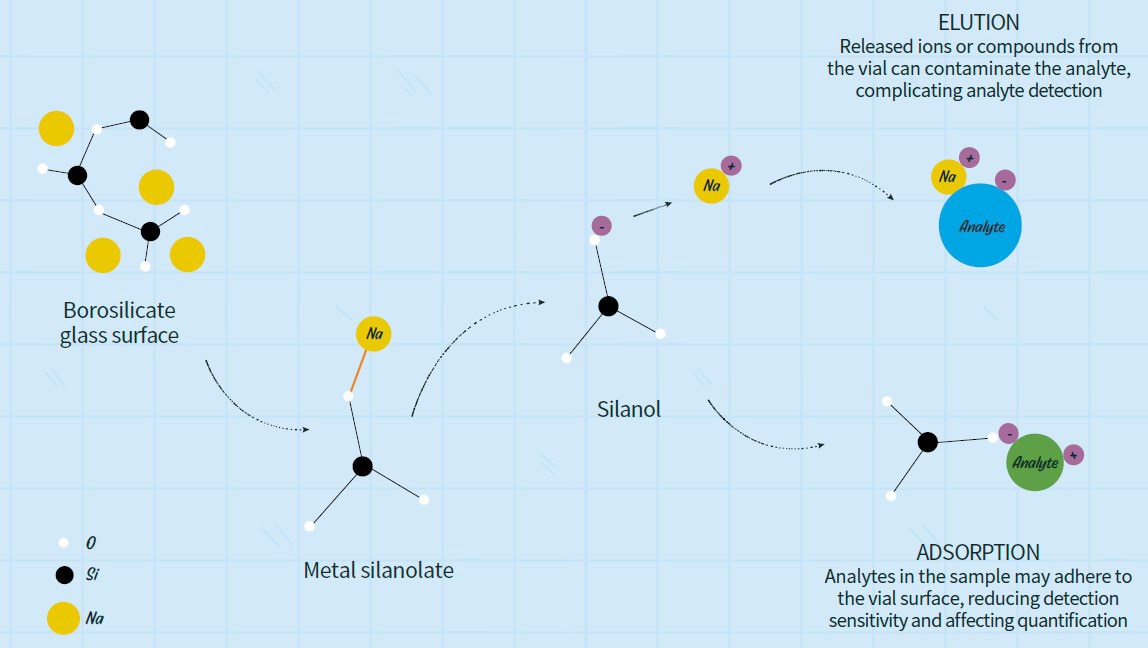
Figure 1. Schematic of interactions between glass vial material and the analyte
Shim-vial S/H glass is a high-quality vial that uses the same material as pharmaceutical filling vials, and undergoes a special low-alkali treatment during molding to reduce the formation of metal silanolate associated with the removal of alkali metals. Shim-vial H glass, in particular, reduces sodium leaching to an absolute minimum, making it well suited for high-sensitivity analysis.
The results of measuring the amount of Na leached from the inner surface of various glass vials are shown in Figure 2. For measurement, pure water was dispensed into vials, pretreated according to USP660, and analyzed by atomic absorption spectrophotometer.
It was confirmed that Shim-vial S/H glass releases a smaller amount of Na into aqueous solution compared with other vials.
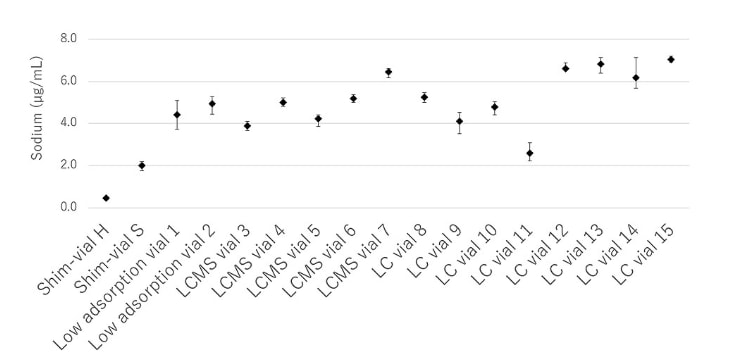
Figure 2. Comparison of Na leaching amounts
In LC/MS analyses in particular, Na dissociated from the vial inner surface can react with ionized analytes, and Na adducts may be detected.
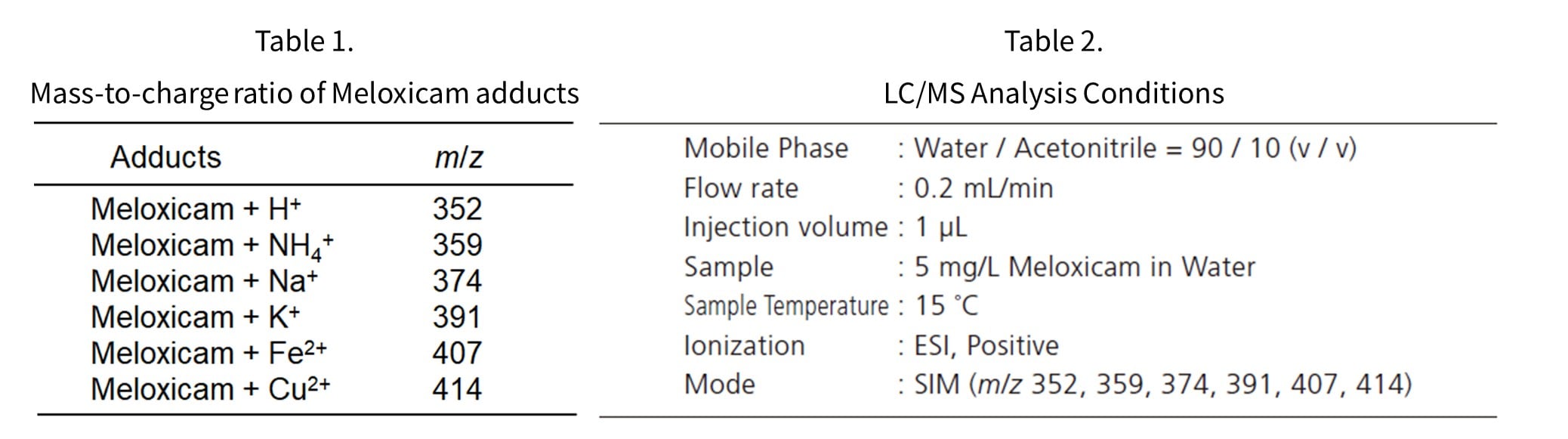
An aqueous solution of 5 mg/L meloxicam was dispensed into each glass vial and left standing for 48 hours before adducts were measured by LC/MS. Figure 3 shows the percentage of each adduct when the sum of the adduct peak areas listed in Table 1 is set to 100%. Analytical conditions are summarized in Table 2.
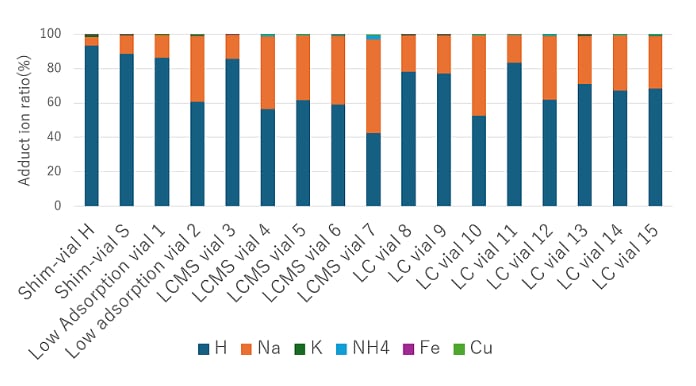
Figure 3. Distribution of added ions by vial difference
Shim-vial H showed the highest proportion of H adducts and is less affected by metal ions originating from the vial. However, some vials were observed in which Na adducts accounted for more than 50%. In particular, in LC/MS quantification targeting H adduct, the formation of Na adducts can reduce sensitivity because only a portion of the H adduct signal contributes to the measurement.
- References
- Fundamentals of Inorganic Glasses, 4 (1994)
- U.S. Pharmacopoeia 41–NF36, 2017 General Chapter <1660>
- npj Materials Degradation 5, 15 (2021)
- Journal of Pharmaceutical and Biomedical Analysis, 213 (2022)
2. Adsorption of the analyte onto the vial
Adsorption of samples to the vial can affect analytical results when analytes bind to adsorption sites on the vial surface. This effect is not noticeable at sufficiently high analyte concentrations, but it tends to become more evident as concentrations decrease.
Specifically, this may lead to:
(1) loss of linearity (the correlation coefficient (r) falls well below 1.000),
(2) poor accuracy (low recoveries), and
(3) poor precision (high variability).
For example, if adsorption phenomena are discovered in vials after optimal separation conditions have been established, a comprehensive revalidation of the analytical method may be required. Therefore, to ensure both development efficiency and method reliability, it is important to identify and understand the adsorption phenomena throughout the entire process from sample preparation to measurement.
As a concrete example, an aqueous solution of chlorhexidine (1 mg/L), a basic compound, was added to various vials, and the recovery of chlorhexidine was determined by LC analysis after 24 and 72 hours. Figure 4 shows the recoveries at each time point, and Table 3 lists the analytical conditions. The results confirmed differences among vials in adsorption behavior, both in recovery and in changes over time.
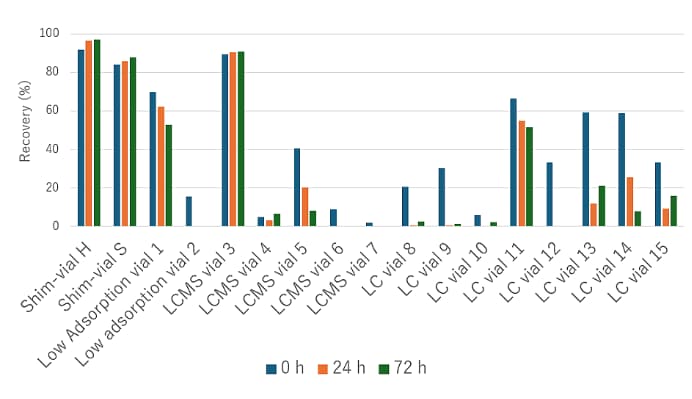
Figure 4. Recovery rate of Chlorhexidine
- Note:
- Recovery is shown for each vial type, with the chlorhexidine peak area measured in polypropylene vials set to 100%.
- Time-course recoveries were obtained by reinjecting samples from the same vial.
- In some vials, effects due to solvent evaporation were observed.
Table 3. HPLC analysis conditions

This adsorption phenomenon occurs when a basic compound is dispensed into a vial, and the basic compound is adsorbed to the silanol on the inner surface of the glass through ion exchange. This adsorption proceeds very quickly immediately after dispensing and then continues more gradually.
Related Products
Shim-vial™ H glass, S glass
Vials are extensively used in an analysis. Accordingly, they are likely to be chosen with a focus on cost. However, if problems with cracks or contamination occur, this can interfere with the analysis or have an impact on data reliability. Shimadzu provides high-quality vials that thoroughly eliminate these risks by visually inspecting each vial, allowing them to be used with confidence.







