TOC in Public Drinking Water
Basics of Total Organic Carbon Analysis
Drinking Water Quality Testing: The Role of TOC Analysis
In which countries is it safe to drink tap water?
Water is essential for our daily lives.
In Japan, we take it for granted that it is safe to drink the water that flows out of our faucets at home, but what about in other countries?
Actually, Japan is among only 9 countries where it is safe to drink tap water.
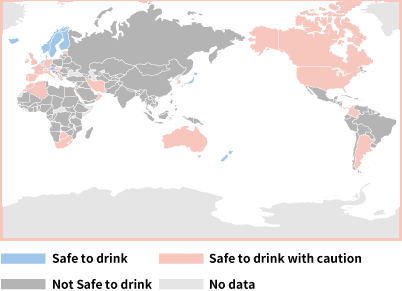
• Source: Japan’s Ministry of Land, Infrastructure, Transport and Tourism “2025 Status of Water Resources in Japan, Ch. 7 International Water Resource Initiatives”
Why are there so many countries with unsafe tap water?
Drinking water quality is often compromised by two primary factors:
- Arid Climates: Dry regions struggle to secure stable water sources as rivers and lakes frequently dry up.
- Infrastructure Costs: In large countries like the United States or China, the cost of building and maintaining a nationwide public water supply system is massive.
Why is tap water safe in Japan?
Due to high rainfall levels, Japan is blessed with many rivers, lakes, and other potential water supply sources. Japan is also a small country that can relatively easily build infrastructure for supplying public water and other needs. However, to ensure safe and good-tasting water, Japan also purifies public drinking water and conducts regular water quality inspections before water is delivered to homes.
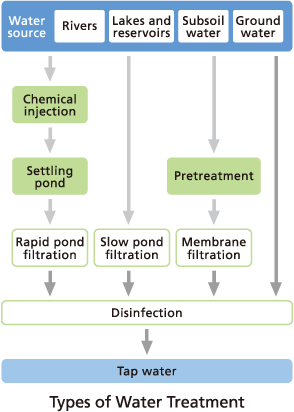
What are the main methods of water purification?
Water purification is the process of removing suspended particles, microorganisms, and chemical contaminants from a water supply. In Japan, four main methods are used:
- Rapid Filtration: Uses chemical coagulation to clump particles followed by sand filtration. It is ideal for high-turbidity water.
- Slow Filtration: Relies on a biofiltration membrane (microorganisms on sand) to naturally remediate water at a slower flow rate.
- Membrane Filtration: Uses microfiltration or ultrafiltration membranes to physically block contaminants.
- Disinfection: Uses chlorine to eliminate microorganisms, typically used for high-quality groundwater sources.
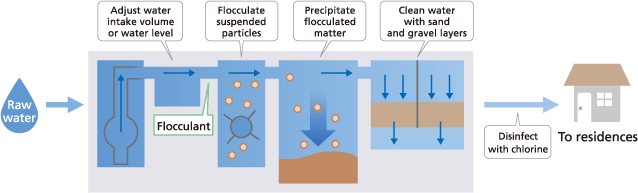
(1) Rapid filtration
This method cleans water by flocculating and precipitating small suspended particles, bacteria, and other substances using chemicals, and then filtering the supernatant through a sand layer in a filtration pond. This method is especially suitable for treating water from rivers, lakes, and other sources with relatively high turbidity levels and is the most commonly used method.
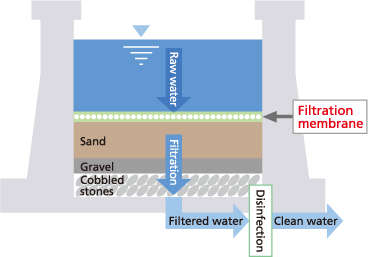
(2) Slow filtration
This method cleans water by passing it slowly through a sand layer in a filtration pond, where microorganisms propagated on the surface of the sand layer (biofiltration membrane) remediate the water. It is referred to as “slow filtration” because the flowrate through the filtration pond is slower than with rapid filtration. It is best suited to treating water sources with relatively good water quality and minimal variations in water quality.
(3) Membrane filtration
This method removes suspended particles, microorganisms, and other substances from water by filtering them through a microfiltration or ultrafiltration membrane.
• Microfiltration and ultrafiltration membranes have different pore sizes or numbers of layers.
(4) Disinfection
This method uses chlorine to only disinfect water obtained from groundwater water sources with good water quality.
Thus, safe tap water is supplied by using a water treatment method appropriate for the water quality of the given river, lake, groundwater, or other water source.
How is drinking water quality testing regulated?
Public drinking water is supplied by using a water treatment based on the water quality of the given river, lake, groundwater, or other water source. However, the water quality of public drinking water can vary due to changes in the water quality or usage rate of the river or lake.
Therefore, it is important to regularly inspect the safety of treated water.
In Japan, the Water Supply Act specifies water quality standards by law.
Japan’s Water Supply Act
In essence, the purpose of the Water Supply Act is to improve the lives of Japan’s citizens by creating public water supply systems throughout Japan that can cheaply deliver large quantities of clean water. However, what does “clean water” really mean?
To address that question, the Water Supply Act specifies water quality standards. Those water quality standards specify 51 types of inspection criteria, with water system contractors regularly inspecting public water systems to make sure public water supplies satisfy those criteria. That means only safe drinking water that passes those strict inspection standards is delivered to homes.
Inspecting Water Quality
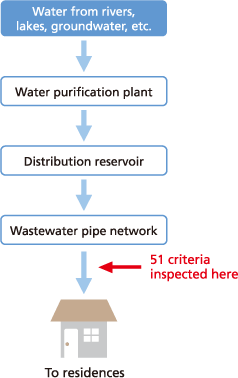
Water from rivers or lakes is treated and supplied from the faucets in our homes as public drinking water according to the process illustrated above. The fact that the 51 types of inspection criteria specified by the Water Supply Act are checked immediately prior to delivery to our homes indicates their importance.
Supplying Water that is Both Safe and Good-Tasting
Furthermore, in Japan, due to the increasing consumption of bottled mineral water, increasingly widespread use of residential water purification systems, and other factors, there is growing interest in water that is not only safe but also good-tasting.
Given such circumstances, to supply safe and good-tasting water, some prefectures have specified water quality goals for the taste of water that are separate from the inspection standards for the 51 criteria specified by the Water Supply Act.
| Inspection Parameter | Description |
|---|---|
| Evaporation Residues | The residues that remain after water is evaporated mainly consist of minerals. Water that contains large amounts of such minerals can taste bitter or astringent, whereas water that contains moderate amounts tend to have a smooth rich taste. |
| Hardness | Hardness indicates the calcium or magnesium content, which are the main mineral components in water. An appropriate level of hardness components is a requirement for good-tasting water. Water with low hardness is referred to as “soft” water and has no distinctive flavor. In contrast, water with high hardness is referred to as “hard” water and has a more persistent flavor, which some people prefer but some do not like. |
| Free Carbon Dioxide | In water, free carbon dioxide refers to dissolved carbon dioxide gas that gives water a crisp sensation, but too much can be overstimulating and reduces smoothness. |
| Potassium Permanganate Consumption | This is an index for organic matter in water. Higher concentrations can result in a bitter taste. |
| Odor Intensity | This indicates the intensity level of odors in water (regardless of the odor type). Moldy odors, algae odors, or other unpleasant odors from water can make water taste bad. |
| Residual Chlorine | This refers to residues of chlorine used to disinfect public drinking water. For public health reasons, public drinking water is required to contain at least 0.1 gm/L of residual chlorine, but if the residual chlorine concentration is too high, it can cause a chlorine bleach odor. |
| Water Temperature | Cold water physiologically tastes better. In addition, chlorine and other odors are less noticeable in cold water, which makes the water taste better. |
Source: Good-Tasting Water Research Committee, “Good-Tasting Water,” Journal of Japan Water Works Association, Vol. 54, No. 5 (1985)
The water treatment and water quality inspection practices described above have resulted in Japan being one of the few countries in the world with tap water that is safe to drink. However, achieving that requires sophisticated technology and high costs. For countries with a small land area, like Japan, it can be relatively easy to build the infrastructure, but for countries with large land areas, infrastructure can require massive amounts of time and expense. Many developing countries have regions without access to public water supply systems.
Even in Japan, if rivers, lakes, and other sources for public water systems become more polluted, the current water treatment infrastructure may not be adequate for ensuring safe and good-tasting water. Therefore, it is important to value safe and good-tasting water as something precious, rather than taking it for granted.
What is TOC in drinking water quality testing?
Total Organic Carbon (TOC) is an index of organic matter that measures the total amount of carbon found in organic compounds within a water sample. In 2005, TOC was introduced as a new water quality standard, replacing older methods like potassium permanganate consumption.
Advantages of TOC over traditional methods:
- Oxidation Efficiency: Enables oxidation of nearly 100% of organic matter.
- Consistency: Unlike manual titration, instrumental TOC analysis does not vary between individuals.
- Speed: Results are available in only a few minutes, allowing for real-time monitoring.
Why is TOC measurement essential for drinking water safety?
- Confirming Safety (DBP Prevention): Organic matter reacts with disinfectants like chlorine to generate harmful Disinfection By-products (DBPs). Measuring TOC is a critical index for identifying these risks.
- Managing Taste: TOC levels directly affect the palatability of tap water. High levels can cause medicinal or bitter tastes.
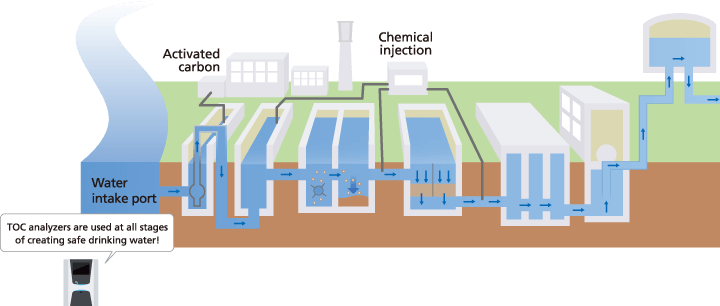
- Water Treatment Management: By measuring TOC at each process step, plants can confirm that filtration and oxidation stages are functioning correctly.
- Cost Optimization: Adjusting chemical dosing based on real-time TOC values can significantly reduce water treatment costs.
FAQ
What is the most important parameter in drinking water quality testing?
While all 51 criteria in the Water Supply Act are important, Total Organic Carbon (TOC) is a primary safety index. It measures the total amount of organic matter, which directly influences the formation of harmful disinfection by-products and determines the overall safety and taste of the water.
Why is TOC measured instead of Potassium Permanganate Consumption?
TOC analysis has replaced potassium permanganate consumption because it is more accurate and consistent. While potassium permanganate consumption results can vary based on the person performing the test, TOC is an instrumental analysis that oxidizes nearly 100% of organic matter, providing reliable data in just a few minutes.
How does organic matter affect the taste of tap water?
Organic matter, measured as TOC, can cause a bitter or medicinal taste and unpleasant odors (such as moldy or algae smells). High levels of TOC can also lead to yellow-to-brown discoloration, which is why monitoring organic levels is essential for supplying "good-tasting" water.
Can TOC testing reduce water treatment costs?
Yes. By implementing TOC measurement at each step of the purification process, water treatment plants can practice data-driven chemical dosing. Instead of using a fixed amount of chlorine or coagulants, operators adjust the dosage based on actual organic loads, significantly reducing chemical waste and operational costs.





