What is “TOC”?
Basics of Total Organic Carbon Analysis
What is TOC (Total Organic Carbon)?
Total organic carbon (TOC) refers to the total amount of carbon contained in organic matter in water. The TOC value is used as a representative water quality index that indicates water cleanliness.
What is the measurement principle of TOC?
TOC is measured by oxidizing organic matter into carbon dioxide (CO2) and detecting the amount of CO2 generated. The measured CO2 concentration corresponds to the total amount of organic carbon present in the sample.
Why is TOC preferred over BOD and COD?
Due to the large number of possible organic substances, biochemical oxygen demand (BOD), chemical oxygen demand (COD), and potassium permanganate consumption tests were traditionally used as indices for collective measurements of all organic substances, regardless of type.
- Precision: Traditional tests (BOD/COD) rely on microorganisms or oxidizing agents that decompose organic substances at different rates, often leading to inconsistent results.
- Speed & Accuracy: TOC measurements are not prone to interference from coexisting substances and provide a more comprehensive measurement of all carbon atoms.
- Versatility: TOC is used across diverse fields, including:
○ Regulating public drinking water safety.
○ Pharmaceutical ultra-pure water monitoring.
○ Environmental research in rivers and soil.
○ Managing industrial factory effluents.
How is TOC classified?
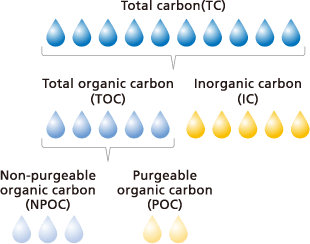
The total amount of all carbon present in water is referred to as “total carbon” (TC). To understand the specific makeup of a sample, TC is categorized into the following hierarchies:
What are the different types of carbon?
- Total Carbon (TC): The sum of all carbon in the sample.
- Inorganic Carbon (IC): Carbonate ions, bicarbonate ions, and dissolved CO2.
- Total Organic Carbon (TOC): The organic fraction, further divided into:
○ NPOC (Non-Purgeable Organic Carbon): Non-volatile organic carbon.
○ POC (Purgeable Organic Carbon): Volatile organic carbon.
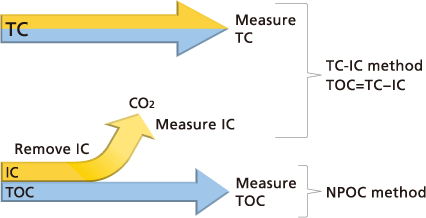
What TOC measurement methods are used?
The following two methods are used to determine the TOC present in water.
TC − IC Method
TOC is calculated by subtracting inorganic carbon (IC) from total carbon (TC).
TOC = TC − IC
NPOC Method
TOC is determined by measuring total carbon after inorganic carbon has been removed from the sample.
TOC = TC
How is inorganic carbon (IC) measured?
Inorganic carbon (IC) is measured by acidifying the sample, which converts carbonate and bicarbonate ions into CO2. The generated CO2 is then detected to determine the IC concentration.
For TOC measurement, IC refers to the total quantity of inorganic carbon contained in water (where CO2 indicates dissolved CO2, HCO3- bicarbonate ions, and CO32- carbonate ions). The quantities of dissolved CO2, bicarbonate ions, and carbonate ions in water are kept in an equilibrium that depends on the pH level of the water, according to the expression below.
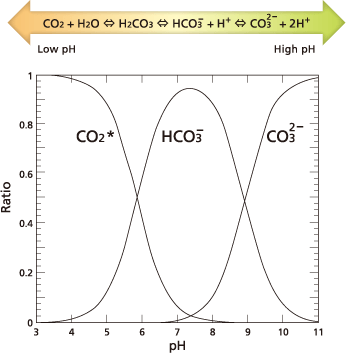
If pH decreases, the equilibrium moves to the left in the expression above. At a pH of 3 or lower, almost all IC becomes dissolved CO2.
Based on that principle, IC is measured by adding acid to lower the sample pH below 3 and then measuring the CO2 extracted from the sample by bubbling in a CO2-free gas atmosphere.
When should you use the TC − IC vs. NPOC method?
Both the TC − IC method and the NPOC method are for measuring TOC, but which of the measurement methods to use is determined based on sample characteristics.
For samples with low IC concentration levels, such as public drinking water or purified water, the NPOC method is used because the TC − IC method is prone to measurement error that could result in lower measurement accuracy.
On the other hand, for samples with large amounts of volatile organic compounds or samples that are prone to foaming, for example, the TC − IC method is used because the NPOC method can result in loss of volatile organic compounds from samples during the bubbling in a CO2-free gas pretreatment step or the NPOC method can prevent accurate sample quantity measurements due to foaming.
What are the methods for TOC oxidation?
To measure total organic carbon, the organic matter must first be oxidized into CO2. The two primary methods are Combustion Oxidation and Wet Oxidation.
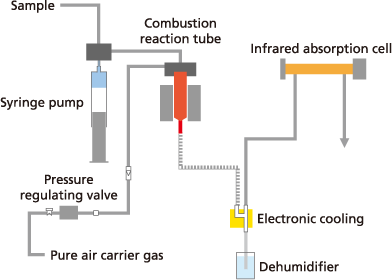
What is the Combustion Oxidation Method?
The combustion oxidation method involves injecting a sample into a high-temperature furnace (typically 650 to 1,200 °C) to incinerate all organic carbon.
- Key Advantage: It efficiently oxidizes "tough" organic matter, including insoluble, macromolecular, or persistent organic substances.
- Best For: Environmental water, factory effluent, and samples containing suspended solids.
- Reagent-Free: Because it relies on heat, this method requires no chemical reagents for pretreatment.
Due to the simplicity of using heat/combustion as the principle for oxidation, the method requires no reagents for pretreatment or posttreatment processes.
What is the Wet Oxidation Method?
The wet oxidation method uses chemical oxidizing agents (often assisted by UV light or heat up to 100 °C) to decompose organic matter into CO2.
- Advantage: Ideal for high-sensitivity measurements in very clean water samples.
- Limitation: It has a weaker oxidative power compared to combustion. It often results in lower carbon recovery rates for particulate organic matter or persistent chemical substances.
What is the history of TOC analyzer innovation at Shimadzu?
Since releasing its first analyzer in 1972, Shimadzu has pioneered the field of Total Organic Carbon (TOC) analysis. Today, these instruments are essential for laboratory quality control, pharmaceutical purified water management, and environmental wastewater monitoring.
Key Milestones in TOC Development:
- 1972: Released the TOC-100 (online) and TOC-10 (laboratory), the first systems designed for the Water Pollution Prevention Act.
- 1983: Developed the TOC-500, the world’s first analyzer with 680 °C combustion catalytic oxidation, significantly improving maintenance and catalyst life.
- 1989: Launched the TOC-5000, expanding measurement sensitivity from ppm to ppb levels, enabling use in ultrapure water applications.
- 2000: Introduced the TOC-V series, adding functionality for solid samples, Purgeable Organic Carbon (POC), and Total Nitrogen (TN).
- 2011–Present: The TOC-L series continues to lead the field in TOC analysis and carbon-neutrality research.
Modern Applications of TOC Analysis: Initially used for environmental water and industrial effluent, TOC performance now covers:
- Pharmaceuticals: Ensuring compliance for purified and ultrapure water.
- Public Safety: Managing drinking water quality standards.
- Sustainability: Researching carbon-neutrality and global environmental protection.
Began development of the first TOC
TOC-10
TOC-10A

TOC-10B

TOC-500
Establishment of a catalytic combustion system at 680°C (low temperature).
Be the basis for subsequent combustion TOC analysis.

TOC-5000
Adoption of automatic liquid delivery by syringe pump.
Fully automatic measurement on a single device.

TOC-5000A
Bestseller.
Enable PC control.

TOC-V
Highly sensitive and multifunctional Total Nitrogen Unit.
First lineup of wet model.

TOC-L
Latest model.
Usability improvements.
Culmination of Shimadzu Lab TOC analysis.

FAQ
What does a high TOC level indicate in water?
A high Total Organic Carbon (TOC) level indicates the presence of organic contaminants, such as microorganisms, industrial chemicals, or decaying matter. In water treatment, high TOC is a critical warning sign because organic matter can react with disinfectants to form harmful by-products and promote bacterial regrowth in piping systems.
What is the difference between TOC and TC?
Total Carbon (TC) is the sum of all carbon atoms in a sample. It is divided into two categories: Total Organic Carbon (TOC) and Inorganic Carbon (IC). The relationship is expressed by the formula: TOC = TC - IC
Why is TOC measurement important in the pharmaceutical industry?
In the pharmaceutical sector, TOC is a mandatory quality index for Purified Water (PW) and Water for Injection (WFI). High TOC levels can interfere with chemical synthesis, reduce the shelf life of drugs, and support the growth of endotoxins, making continuous TOC monitoring essential for regulatory compliance.
How does pH affect TOC measurement?
The pH level determines the chemical state of inorganic carbon. By adding acid to lower the sample pH to 3 or below, carbonates and bicarbonates are converted into dissolved CO2. This allows the CO2 to be stripped away (in the NPOC method) or measured separately (in the TC − IC method) to ensure the final reading represents only organic carbon.
When should you use the NPOC method instead of the TC − IC method?
The NPOC method is best for samples with high inorganic carbon but low organic content (like drinking water) to ensure accuracy. However, the TC − IC method should be used if the sample contains volatile organic compounds (VOCs) or is prone to foaming, as the gas-bubbling step in the NPOC method would cause these organic carbons to be lost before they can be measured.





