Nexera lite inert - Features
High Performance Liquid Chromatograph
-
Improved Data Reliability by Preventing System Corrosion from Halogenic Salt
-
Superior Retention Time Stability by Highly Accurate Solvent Delivery
-
Real-Time Monitoring of Mobile Phase pH
-
Analysis of Antibody Drugs by Gel Filtration Chromatography
-
Analysis of Structural Changes in Proteins Using a Fluorescence Detector
-
Target Compounds Recovered with a Fraction Collector for the Next Analysis Step
-
Purifying and Selectively Recovering Fluorescently Labeled Antibodies
Improved Data Reliability by Preventing System Corrosion from Halogenic Salt
For analysis of antibody drugs or proteins in biological samples,a metal-free LC system is required for good repeatability over a long time due to high-concentration, salt-containing mobile phases such as Sodium chloride. With no metal material in the low path, Nexera lite inert provides stable aggregate assay results with good repeatability.
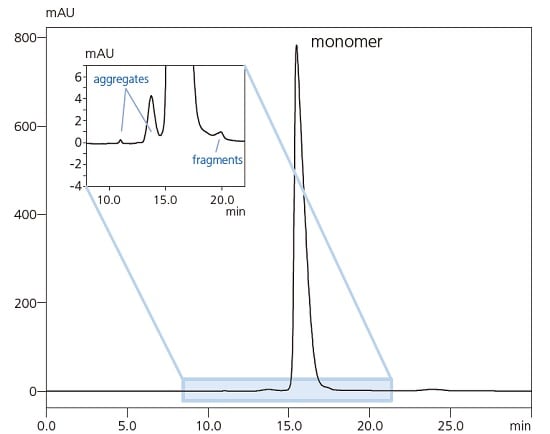
-

- Monoclonal IgG antibody aggregate analysis by gel filtration chromatography
- Corrosion resistance test results of pump heads
Superior Retention Time Stability by Highly Accurate Solvent Delivery
With the outstanding flow-rate stability, the Nexera lite inert provides excellent retention time repeatability.
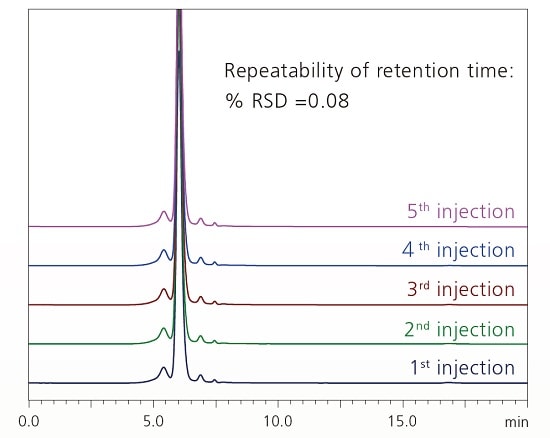
Analysis of the aggregates in the antibody drug (n=5)
Real-Time Monitoring of Mobile Phase pH
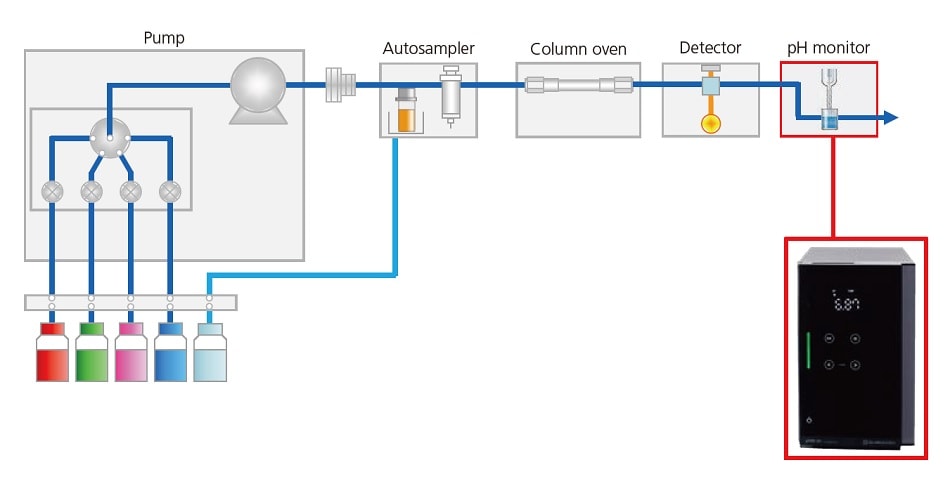
In ion exchange or size exclusion chromatography, the pH of the mobile phase sometimes affects the separation of compounds. The pH Monitor, pHM-40, continuously monitors and records the pH of the mobile phase. The pH and detector chromatograms are saved within the same data file and can be overlapped, ensuring efficient data traceability.
Analysis of Antibody Drugs by Gel Filtration Chromatography
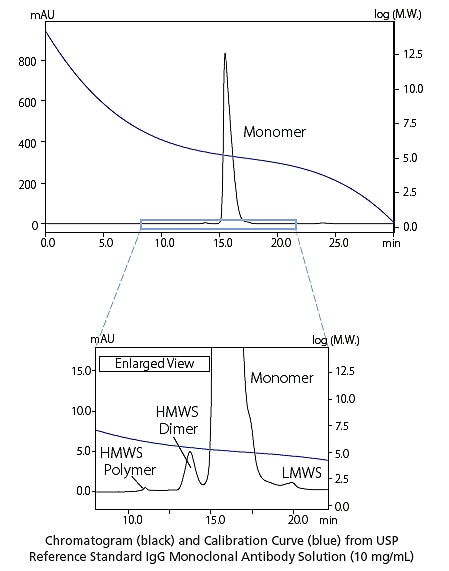
Antibody aggregates indicate the immunogenic potential. In addition, aggregates can result in lower purification efficiency and lower production quantities of antibodies, which are the main active ingredient in antibody drugs. Therefore, antibody drug aggregate analysis is an important process in the manufacturing and quality control of biopharmaceuticals. Using a Nexera lite inert system ensures reliable data can be acquired without worrying about protein adsorption or the risk of corrosion by high salt concentrations.
Analysis of Structural Changes in Proteins Using a Fluorescence Detector
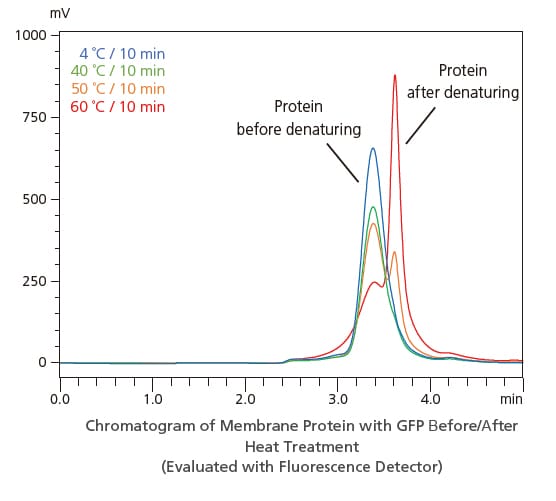
In addition to a general-purpose absorbance detector, Nexera lite inert systems support using a metal-free inert cell with a fluorescence detector, expanding their use to a wider range of applications. The figure on the right is from an example of using gel filtration chromatography to evaluate the molecular weight distribution of heat-treated membrane proteins that contain green fluorescent protein (GFP). It shows that structural changes due to the heat treatment temperature can be detected by fluorescence detection.
Target Compounds Recovered with a Fraction Collector for the Next Analysis Step
Preparative separation of target peaks is possible by connecting an FRC-10A fraction collector to the Nexera lite inert system. The FRC-10A includes no metal materials in low channels, which allows the configuration of systems that are fully inert through to target recovery. In addition, the FRC-10A is compatible with 1.5 mL tubes, 15 mL centrifuge tubes, and 96-well DWPs, so targets can be collected in containers appropriate for subsequent process steps.

Purifying and Selectively Recovering Fluorescently Labeled Antibodies
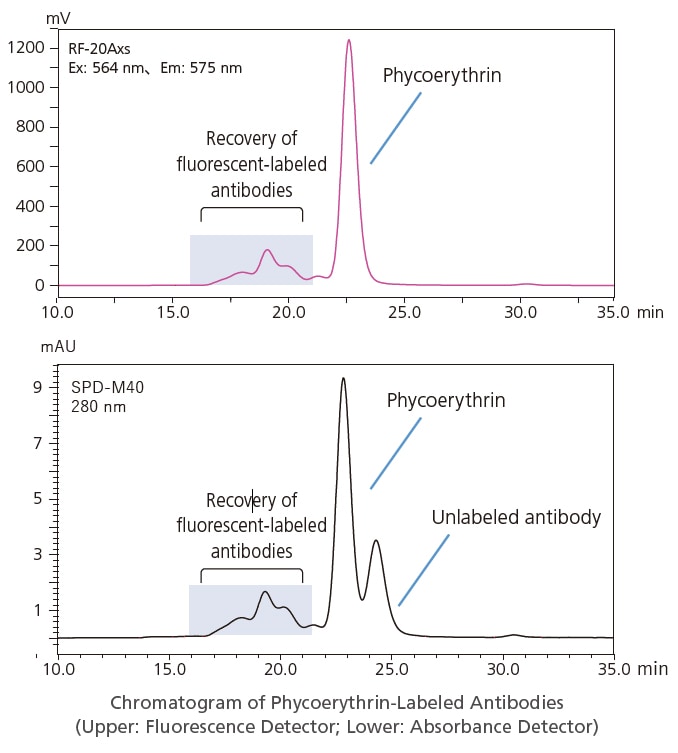
Using a combination of multiple detectors and fraction collectors offers the feexibility to purify samples based on the specific characteristics of target compounds. For example, by using both an absorbance detector and fluorescence detector, antibodies with and without fluorescent labeling can be monitored at the same time. Furthermore, by using an FRC-10A fraction collector to ensure reliable preparative separation of only target components, fluorescently labeled antibodies can be used directly in the next process step.


