Example: For a 30 mg daily...
Co-Sense for Impurities
Liquid Chromatograph with online Sample Preparation

The FDA draft guidance, "Genotoxic and Carcinogenic Impurities in Drug Substances and Products: Recommended Approaches" prescribes permitted limits for genotoxic impurities in drug substances. These impurities demand more sensitive trace-level analysis than normal impurities. Generally, mass spectrometry methods, such as GC/MS or LC/MS, are used for the high-sensitivity analysis of impurities. However, there is increasing demand for the establishment of a high-sensitivity quantitation method using an absorbance or other conventional detector that is easy to operate and can easily apply existing LC analysis conditions.
Features
-
Impurity intake restricted to 1.5 µg/day max. when taking a drug over a long period (12 months or more)
Example: For a 30 mg daily... -
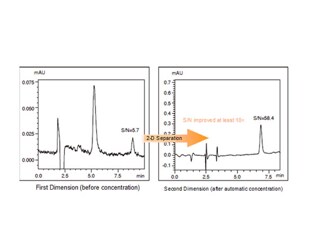
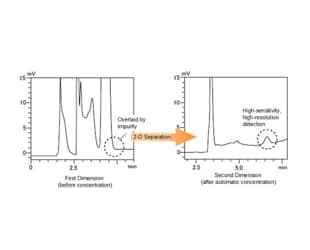
The Co-Sense for Impurities System achieves approximately 10 to 20 times higher sensitivity than 1-D separation only by...
-
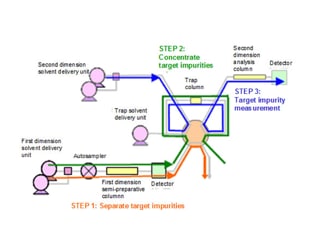
Using a 1-D and 2-D column with different retention characteristics, or using different mobile phase compositions, permits reliable separation and...
-
Significant knowledge and experience are generally required to set the analytical conditions and clean the flow lines of a 2-D separation system. However...
News / Events
-
Shimadzu has released the Nexera X4 Ultra High Performance Liquid Chromatograph.
Expanding on technologies cultivated with previous Nexera series models, the next-generation Nexera X4 ultra high performance liquid chromatograph (UHPLC) achieves an incomparable level of analytical performance. The lowest degree of peak broadening* in the industry results in ultra-sharp peaks and outstanding separation performance while cutting-edge fluid control technology provides exceptional solvent delivery consistency, even for fast analysis under ultra-high-pressure conditions, and highly reliable results. Engineered with precision, the high-end Nexera X4 UHPLC offers new value for all laboratories that demand maximum performance.
-
Nexera IC Ion Chromatograph has been released.
The Nexera IC ion chromatograph combines a compact footprint with uncompromising performance, enabling anyone to perform analysis effortlessly and efficiently.
-
PL-40, Automation-Compatible Plate Loader for LC/LC-MS Systems has been released
The PL-40 solves for LC/LC-MS automation, it seamlessly loads plates of pretreated samples into an LC/LC-MS system, enabling a fully automated workflow, from pretreatment to analysis.
-
New Technical Report is available, Optimization of Supercritical Fluid Extraction Parameters for Vitamins D2, D3, and K1 from Pharmaceutical Preparations
New Technical Report is available, Optimization of Supercritical Fluid Extraction Parameters for Vitamins D2, D3, and K1 from Pharmaceutical Preparations
-
New Tips & Tricks is available, Automated Dilution and Preparation of Standard and Sample Solutions for Analysis
-
Shimadzu Corporation has released LabSolutions Detect, a software with AI Functionality to support Anomaly Detection for Liquid Chromatographs (LC).
LabSolutions Detect transforms your LC data review process by visualizing differences between accumulated reference data and daily sample data.