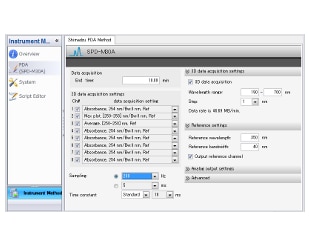
Shimadzu LC Driver for Chromeleon™ 7

Control of Shimadzu’s Nexera and i-Series by Thermo Scientific™ Dionex™ Chromeleon™ Chromatography Data System (CDS) Software (hereafter referred to as "Chromeleon™") 7 is now available through a collaboration between Thermo Fisher Scientific Inc. and Shimadzu utilizing the "Chromeleon Driver Development Kit" for multi-vendor hardware control.
*Chromeleon™ is a registered trademarks of Thermo Fisher Scientific Inc.
Features
-
Shimadzu LC control parameters are set through the Chromeleon Instrument Method Editor embedded in the Chromeleon software. These parameters are stored and handled as standard Chromeleon Instrument methods so that the instruments are directly controlled from Chromeleon...
-
In addition to seamless system control, the detector signal is digitally transmitted to the data system. This eliminates the necessity to add an A/D converter for analogue data acquisition, making system validation easier and more robust.
-
Full system control uses Chromeleon Instrument Method, and data processing is integrated with Chromeleon software so that the audit trail functions of Chromeleon software are fully utilized. This gives you full confidence in the compliance of the system to regulations such as GLP/GMP and US FDA 21 CFR Part 11.
News / Events
-
LabSolutions Insight Profiler, Non-Targeted Analysis Software for LC/QTOF Data has been released
LabSolutions Insight Profiler is a unified, end-to-end workflow for LC/QTOF data processing — with a "single-click" approach. Insight Profiler is purpose-built for non-targeted analysis in suspect screening, unknown identification, and metabolomics across complex sample sets.
-
Shimadzu has released the System Linkage Option
Data obtained with LabSolutions can be output in a variety of formats, and can be coordinated with a host system such as a laboratory information management system (LIMS).
-
LabSolutions Insight Environmental Option is now available
LabSolutions Insight Environmental Option is a software for the environmental regulatory field. In the environmental regulatory field, the quality control (hereinafter QC) requirements prescribed by the United State Environmental Protection Agency (EPA) must be satisfied.
-
Shimadzu has released the LabSolutions i-QLinks.
LabSolutions™ i-QLinks provides integrated control over the quality testing operations of an analytical laboratory, including the preparation of test plans and instructions, the incorporation of test results from analytical instruments such as HPLC, the automatic preparation of test reports from the incorporated test results, and the management of the quality test progress.
-
Shimadzu has released the LabSolutions Sync
Synchronizes Third-Party Software for Pretreatment Units with Shimadzu LC and LC-MS
-
Efficient Method Development by automated pH Screening with LabSolutions MD
This article describes an example of using LabSolutions MD, a dedicated software for supporting method development, to automate pH screening by varying the mobile phase pH from 2.5 to 8.5 to evaluate the optimal pH level for separating 12 small-molecule drugs.